Reports
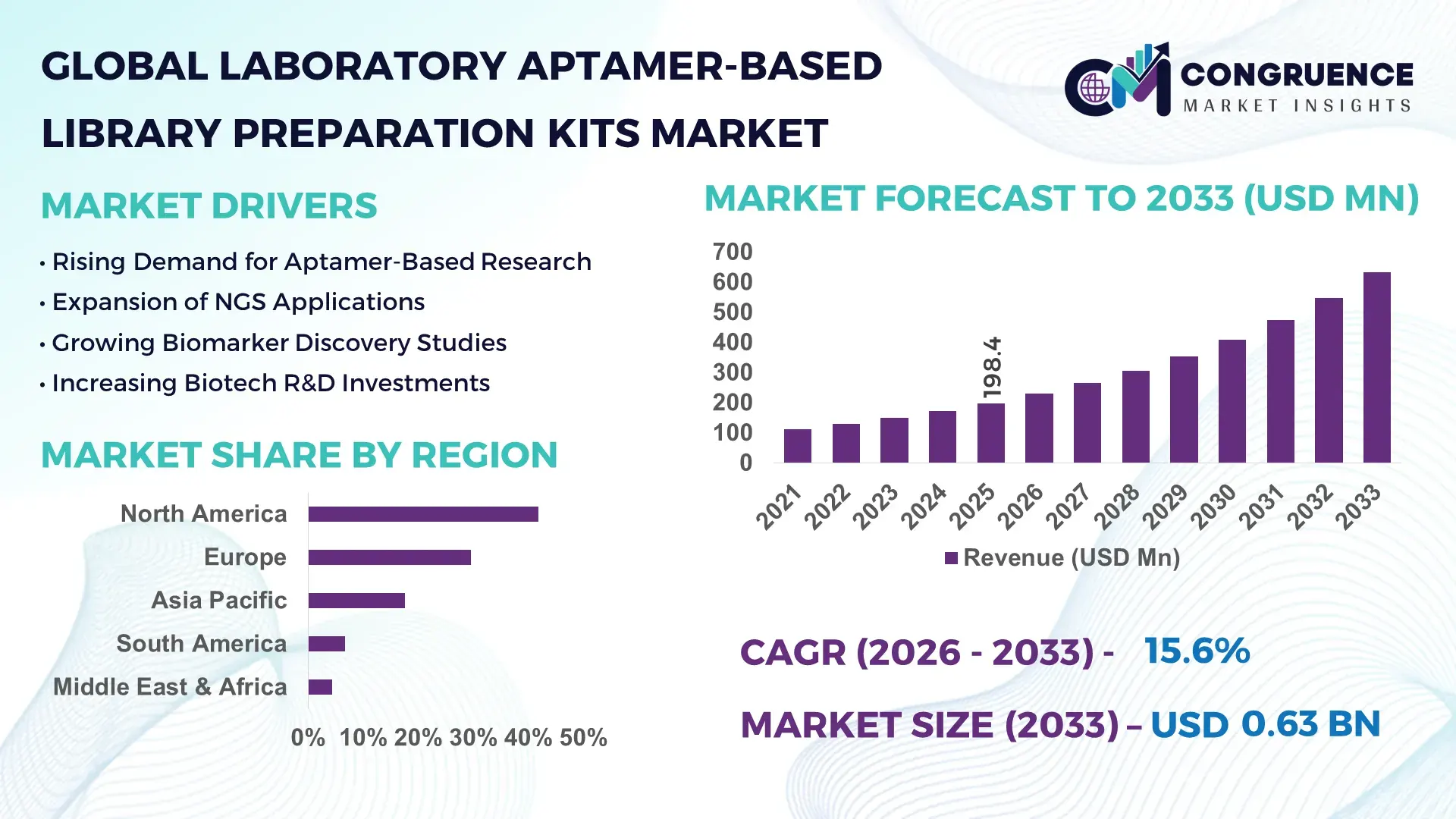
The Global Laboratory Aptamer-Based Library Preparation Kits Market was valued at USD 198.4 Million in 2025 and is anticipated to reach a value of USD 632.7 Million by 2033 expanding at a CAGR of 15.6% between 2026 and 2033, according to an analysis by Congruence Market Insights. The growth is primarily driven by rising adoption of aptamer-based technologies in genomic research, drug discovery pipelines, and high-throughput sequencing workflows across biotechnology laboratories.
The United States remains the most prominent country in the Laboratory Aptamer-Based Library Preparation Kits ecosystem due to strong biotechnology infrastructure, extensive sequencing facilities, and high R&D expenditure. The country accounts for over 40% of global genomic sequencing capacity, supported by more than 2,500 active genomics laboratories and sequencing centers. Federal funding programs allocate over USD 4 billion annually toward genomic and molecular biology research, which directly fuels demand for aptamer library preparation kits. In pharmaceutical R&D, nearly 58% of drug discovery programs incorporate nucleic acid-based screening tools, including aptamer libraries for biomarker identification and targeted therapeutics. Furthermore, more than 65% of large U.S. research institutions have adopted next-generation sequencing workflows that integrate aptamer-based enrichment techniques, highlighting the country’s strong technological advancement and industrial application in molecular diagnostics and proteomics research.
Market Size & Growth: The market stood at USD 198.4 million in 2025 and is projected to reach USD 632.7 million by 2033, expanding at 15.6% CAGR, supported by increasing genomic sequencing adoption and precision medicine initiatives.
Top Growth Drivers: High-throughput sequencing adoption (~68% research labs), biomarker-based drug discovery integration (54% pharmaceutical pipelines), and automation in molecular biology workflows improving efficiency by 35%.
Short-Term Forecast: By 2028, automated library preparation platforms are expected to reduce laboratory processing time by 30% while improving sequencing workflow efficiency by 25%.
Emerging Technologies: AI-assisted aptamer screening, microfluidic-based SELEX automation platforms, and nanopore sequencing integration for rapid nucleic acid library preparation.
Regional Leaders: North America projected at USD 255 million by 2033 with strong genomics funding; Europe expected near USD 185 million driven by collaborative biomedical programs; Asia-Pacific projected around USD 150 million supported by expanding biotechnology manufacturing hubs.
Consumer/End-User Trends: Biotechnology firms and genomic research institutes account for nearly 60% of demand, while clinical diagnostics laboratories show growing adoption for biomarker discovery and targeted therapeutics research.
Pilot or Case Example: In 2024, a genomics research consortium deployed automated aptamer library preparation workflows that improved sequencing throughput by 40% and reduced manual processing errors by 28%.
Competitive Landscape: Illumina leads with roughly 28% share, followed by major participants including Thermo Fisher Scientific, Agilent Technologies, QIAGEN, and New England Biolabs.
Regulatory & ESG Impact: Genomic research guidelines and laboratory safety frameworks are encouraging sustainable reagent usage and 20% reduction in laboratory waste through optimized reagent volumes.
Investment & Funding Patterns: Global biotechnology funding exceeded USD 2.1 billion in research grants and venture investment supporting nucleic acid technologies and sequencing tool development.
Innovation & Future Outlook: Integrated sequencing platforms, AI-driven biomarker discovery, and advanced nucleic acid enrichment technologies are expected to significantly reshape laboratory workflows in genomic research.
Aptamer-based library preparation kits are increasingly utilized across pharmaceutical research (≈42% usage), academic genomics laboratories (≈35%), and clinical diagnostics applications (≈23%). Innovations such as automated SELEX platforms and microfluidic nucleic acid enrichment systems are improving screening efficiency by nearly 30%, while regulatory initiatives supporting precision medicine programs are accelerating adoption. Demand is particularly strong in North America and East Asia, where genomics infrastructure and biotechnology investment continue expanding.
The Laboratory Aptamer-Based Library Preparation Kits Market holds strategic relevance within modern molecular biology and genomic research infrastructure because aptamer-based screening technologies enable highly selective nucleic acid libraries for biomarker discovery, therapeutic target identification, and precision diagnostics. In drug discovery workflows, aptamer libraries provide molecular recognition capabilities that support advanced proteomics and genomic screening. Approximately 54% of pharmaceutical R&D programs now incorporate nucleic acid-based molecular tools, and laboratory automation platforms have increased sample processing throughput by 35–40%.
Technological innovation is rapidly improving the performance of aptamer library preparation kits. For example, microfluidic SELEX platforms deliver nearly 45% faster screening efficiency compared to conventional SELEX protocols, significantly reducing laboratory time for aptamer selection. Similarly, automated next-generation sequencing-integrated library preparation platforms enable laboratories to process up to 96 samples simultaneously, improving experimental reproducibility and reducing manual handling errors.
Regional dynamics show contrasting strengths. North America dominates in sequencing volume, supported by advanced biotechnology infrastructure and extensive R&D funding, while Asia-Pacific leads in adoption expansion with nearly 48% of emerging biotechnology firms integrating high-throughput nucleic acid technologies. Countries such as China, Japan, and South Korea are investing heavily in genomics research laboratories and bio-innovation clusters.
Short-term technological evolution is expected to accelerate. By 2028, AI-driven aptamer discovery algorithms are projected to improve biomarker screening accuracy by nearly 30% while reducing experimental design time by 25%. These systems analyze sequencing data patterns to optimize aptamer candidate selection more efficiently than traditional screening approaches.
Sustainability and compliance are also shaping future strategies. Research institutions are committing to laboratory reagent optimization programs aimed at reducing chemical waste by 20% by 2030, while adopting automated liquid-handling systems that minimize reagent consumption.
A notable micro-scenario illustrates this transformation: in 2024, a U.S. genomics research center integrated AI-assisted aptamer screening software into its sequencing pipeline, achieving a 32% improvement in biomarker identification efficiency while reducing experimental cycles by 18%.
Looking ahead, the Laboratory Aptamer-Based Library Preparation Kits Market is positioned as a critical pillar supporting biotechnology innovation, regulatory compliance, and sustainable laboratory operations, enabling advanced genomic research and next-generation therapeutic discovery worldwide.
The Laboratory Aptamer-Based Library Preparation Kits Market is evolving as genomic research and nucleic acid-based technologies expand across biotechnology, pharmaceutical, and clinical diagnostic applications. The increasing adoption of next-generation sequencing (NGS) technologies, along with advancements in aptamer selection processes such as Systematic Evolution of Ligands by Exponential Enrichment (SELEX), is transforming laboratory workflows. Over 70% of modern molecular biology laboratories now employ high-throughput sequencing tools, creating strong demand for efficient library preparation systems that can support rapid nucleic acid enrichment and biomarker discovery.
Biotechnology companies are integrating aptamer libraries into drug discovery pipelines due to their high specificity toward protein targets. Studies indicate that aptamers demonstrate binding affinities comparable to monoclonal antibodies, making them valuable for diagnostic assays and therapeutic development. Additionally, automation technologies in laboratory environments are significantly improving throughput. Automated liquid-handling systems and robotic sample preparation platforms have increased laboratory productivity by 30–40%, which directly boosts the demand for standardized library preparation kits.
Government funding for genomic research and personalized medicine programs also plays a critical role. Multiple countries are investing heavily in national genome sequencing initiatives, leading to the expansion of genomic laboratories and bioinformatics infrastructure. At the same time, pharmaceutical companies are focusing on targeted drug discovery approaches, where aptamer-based screening helps identify disease biomarkers with higher precision. These dynamics collectively shape the evolving market landscape.
Precision medicine initiatives and large-scale genomic sequencing projects are significantly accelerating the adoption of laboratory aptamer-based library preparation kits. Modern precision medicine approaches rely heavily on molecular profiling of diseases, where aptamer libraries help identify biomarkers with high binding specificity. Nearly 65% of oncology research programs now involve genomic sequencing techniques, which require robust nucleic acid library preparation workflows. Aptamers are particularly valuable in proteomics and biomarker discovery because they can bind selectively to proteins, peptides, and other molecular targets with high affinity. Research institutions are also scaling up sequencing capacity to analyze larger patient datasets. Large genomic laboratories can process thousands of sequencing samples per week, increasing the need for efficient library preparation kits that ensure reproducible results. In addition, pharmaceutical companies are incorporating aptamer-based screening during early drug discovery stages to evaluate molecular targets more effectively. Studies suggest that aptamer-based assays can reduce biomarker discovery timelines by nearly 25%, accelerating therapeutic development. The integration of automated sequencing workflows and advanced molecular biology tools further strengthens demand for standardized laboratory kits across research institutions and biotechnology firms.
Despite the growing adoption of aptamer technologies, the complexity of aptamer selection and validation processes presents a notable restraint for the Laboratory Aptamer-Based Library Preparation Kits Market. The SELEX process, commonly used for aptamer development, often requires multiple rounds of nucleic acid amplification and target screening before identifying high-affinity sequences. Some selection processes require 10–15 iterative cycles, which increases experimental time and laboratory resource consumption. Another challenge lies in the requirement for specialized expertise and equipment. Advanced molecular biology laboratories must employ trained researchers with expertise in nucleic acid chemistry, bioinformatics, and sequencing technologies. Surveys indicate that over 40% of smaller biotechnology startups face operational barriers when implementing advanced aptamer workflows due to limited infrastructure and high equipment costs. Furthermore, compatibility issues between library preparation kits and various sequencing platforms may create operational constraints. Laboratories using different NGS systems often need optimized protocols for reagent compatibility and sample preparation. These technical limitations slow adoption among institutions with limited resources, particularly in emerging biotechnology markets where research infrastructure is still developing.
Biomarker discovery initiatives across oncology, infectious disease research, and neurological disorders present a significant opportunity for the Laboratory Aptamer-Based Library Preparation Kits Market. Healthcare systems worldwide are investing in advanced diagnostic tools capable of detecting diseases at earlier stages. Aptamer-based screening technologies provide high sensitivity in identifying proteins and molecular markers associated with disease progression. Global research programs investigating cancer biomarkers have expanded by more than 35% in the past five years, increasing demand for efficient nucleic acid library preparation kits. Aptamers can bind to complex molecular targets that traditional antibody-based assays may fail to detect, enabling researchers to explore new therapeutic pathways. Additionally, pharmaceutical companies are adopting high-throughput screening platforms capable of analyzing thousands of potential biomarker targets simultaneously. Emerging biotechnology clusters in Asia-Pacific and Europe are investing heavily in genomic infrastructure, including automated sequencing laboratories and bioinformatics platforms. These investments create opportunities for suppliers of aptamer library preparation kits to expand their presence in research institutes, pharmaceutical R&D facilities, and clinical diagnostic laboratories that are seeking more efficient molecular screening solutions.
Regulatory oversight in molecular diagnostics and genomic research laboratories is becoming increasingly stringent, creating operational challenges for the Laboratory Aptamer-Based Library Preparation Kits Market. Regulatory bodies require laboratories to comply with strict quality control, validation, and documentation standards before implementing new molecular testing tools. These compliance requirements often necessitate extensive validation studies to confirm the reliability and reproducibility of aptamer-based assays. Laboratories conducting clinical diagnostic research must follow internationally recognized laboratory accreditation standards, which include strict protocols for reagent quality, contamination control, and experimental reproducibility. Implementing new library preparation kits often requires additional validation procedures to ensure compatibility with sequencing instruments and existing workflows. Compliance processes may increase development timelines and require significant laboratory resources. Additionally, global regulatory agencies are emphasizing traceability and quality assurance for biological reagents, which requires manufacturers to implement advanced quality monitoring systems during production. These regulatory expectations increase operational complexity for suppliers while slowing product deployment timelines in research and clinical environments.
Rapid Automation of Library Preparation Workflows: Automation is transforming molecular biology laboratories, with over 62% of high-throughput genomic laboratories adopting robotic liquid-handling systems for nucleic acid preparation. Automated library preparation platforms can process 96–384 samples simultaneously, reducing manual errors by nearly 30% and increasing laboratory throughput by more than 40%. This trend is particularly prominent in North American and European genomic centers where research facilities handle large-scale sequencing datasets.
Integration of AI-Driven Aptamer Screening Technologies: Artificial intelligence is increasingly used to analyze sequencing data and optimize aptamer selection. Machine learning algorithms can evaluate millions of nucleotide sequences in minutes, improving candidate screening accuracy by 35% compared with traditional computational methods. Approximately 48% of biotechnology research institutions are now experimenting with AI-assisted biomarker discovery platforms integrated with aptamer libraries.
Expansion of High-Throughput Sequencing Facilities: Global genomic sequencing capacity has increased significantly in recent years, with more than 3,000 operational sequencing laboratories worldwide. Large research institutions now operate sequencing platforms capable of generating terabytes of genomic data per week, requiring efficient library preparation solutions. The number of laboratories adopting aptamer-based enrichment methods increased by nearly 28% between 2022 and 2025, reflecting the rising importance of targeted molecular screening.
Rising Role of Aptamers in Therapeutic and Diagnostic Research: Aptamers are gaining traction as alternatives to monoclonal antibodies in therapeutic and diagnostic applications. Studies show that aptamers can achieve binding affinities exceeding 90% specificity for target proteins, making them valuable tools for biomarker detection and drug delivery research. Pharmaceutical companies have expanded aptamer-based research programs by nearly 33% in the past five years, particularly in oncology and infectious disease research.
The Laboratory Aptamer-Based Library Preparation Kits Market is segmented based on type, application, and end-user, reflecting the diverse usage of nucleic acid library preparation tools across genomic research and biotechnology industries. Each segment contributes uniquely to the market landscape depending on laboratory workflow requirements and research objectives. Different types of kits are designed to support various aptamer screening protocols and sequencing technologies, enabling laboratories to select solutions compatible with their experimental design. Applications span drug discovery, biomarker identification, diagnostics research, and proteomics analysis, demonstrating the broad scientific relevance of aptamer-based molecular tools. End-user adoption varies widely among academic research institutes, biotechnology firms, and pharmaceutical companies that utilize high-throughput sequencing workflows. Increasing automation in genomic laboratories, combined with the rising need for precise molecular screening technologies, is influencing how these segments evolve. The segmentation structure provides insights into how laboratories and research organizations deploy aptamer library preparation kits to accelerate genomic analysis and therapeutic discovery.
Laboratory Aptamer-Based Library Preparation Kits are primarily categorized into SELEX-based library preparation kits, automated microfluidic library preparation kits, and NGS-compatible aptamer enrichment kits. Among these, SELEX-based library preparation kits hold the leading position with nearly 46% adoption, as they remain the standard methodology for aptamer discovery and nucleic acid enrichment in molecular biology research. Their widespread use stems from the established reliability of SELEX protocols in generating high-affinity aptamer sequences for protein and biomarker targeting. Automated microfluidic library preparation kits represent the fastest-growing segment, expanding at an estimated 17.8% growth rate due to their ability to streamline complex experimental steps such as nucleic acid amplification and selection cycles. Microfluidic systems can reduce reagent consumption by nearly 40% while improving screening throughput, making them attractive for high-volume research laboratories. NGS-compatible aptamer enrichment kits and other specialized formats collectively account for approximately 32% of the segment, serving laboratories conducting targeted genomic sequencing and advanced biomarker discovery experiments. These kits enable efficient preparation of nucleic acid libraries compatible with high-throughput sequencing instruments.
• In 2024, a major genomics research institute implemented automated microfluidic SELEX systems capable of screening over 1 million aptamer candidates per experiment, accelerating biomarker identification across multiple disease research programs.
Applications of Laboratory Aptamer-Based Library Preparation Kits include drug discovery and biomarker identification, diagnostic research, proteomics studies, and molecular interaction analysis. Among these, drug discovery and biomarker identification account for approximately 44% of total adoption, as aptamers are widely used to identify molecular targets and disease-associated proteins during early therapeutic research. Diagnostic research represents the fastest-growing application segment, expanding at around 16.9% growth, driven by the rising demand for precision diagnostics and molecular screening technologies. Aptamer libraries are increasingly utilized to detect disease biomarkers associated with oncology, cardiovascular disorders, and infectious diseases. Other applications such as proteomics and molecular interaction studies contribute a combined 39% share, supporting research activities focused on protein binding, cellular signaling pathways, and molecular biology experimentation. In terms of adoption patterns, over 52% of biotechnology companies report integrating nucleic acid-based screening technologies into their R&D pipelines, while approximately 41% of genomic laboratories employ aptamer libraries in experimental proteomics research.
• In 2025, a national biomedical research program deployed aptamer-based biomarker screening across more than 120 clinical research laboratories to accelerate early detection studies for chronic diseases.
The primary end-users of Laboratory Aptamer-Based Library Preparation Kits include academic and research institutes, biotechnology companies, pharmaceutical firms, and clinical diagnostic laboratories. Academic and research institutes dominate the segment with approximately 38% adoption, as universities and government-funded laboratories conduct large-scale genomic and molecular biology research requiring high-throughput nucleic acid screening. Biotechnology companies represent the fastest-growing end-user group with an estimated 18.2% growth rate, driven by rapid expansion of biotech startups focused on molecular diagnostics, biomarker discovery, and therapeutic development. Many biotech firms are investing heavily in automated sequencing laboratories and AI-driven data analysis platforms to accelerate research timelines. Pharmaceutical companies and clinical diagnostic laboratories collectively account for around 44% of the segment, using aptamer-based library preparation kits in drug target validation, therapeutic discovery, and molecular diagnostic assay development. Adoption patterns show that nearly 47% of pharmaceutical research programs now utilize advanced sequencing workflows, while over 42% of hospitals in developed healthcare systems are exploring nucleic acid-based molecular diagnostics platforms.
• In 2024, a government-supported genomic research initiative enabled more than 300 university laboratories to integrate automated aptamer library preparation workflows for large-scale biomarker screening projects.
North America accounted for the largest market share at 41.8% in 2025 however, Asia-Pacific is expected to register the fastest growth, expanding at a CAGR of 17.9% between 2026 and 2033.
The regional landscape of the Laboratory Aptamer-Based Library Preparation Kits Market reflects strong research infrastructure, biotechnology investment, and genomic sequencing capacity across major economies. North America maintains its leadership due to a dense network of more than 2,500 genomic research laboratories and sequencing centers, along with advanced biotechnology companies integrating aptamer technologies in molecular diagnostics and drug discovery programs. Europe represents the second-largest regional ecosystem with approximately 29.6% share, supported by collaborative biomedical research initiatives and national genome sequencing programs across countries such as Germany, the United Kingdom, and France. Asia-Pacific is witnessing rapid expansion due to large-scale genomic infrastructure development, with over 1,200 new biotechnology startups established between 2020 and 2025. Meanwhile, South America contributes around 6.7% of global demand, primarily driven by Brazil and Argentina where research institutes are expanding molecular biology capabilities. The Middle East & Africa collectively represent 4.3% of the market, supported by increasing investments in biomedical research laboratories and precision medicine initiatives in countries including the UAE, Saudi Arabia, and South Africa.
North America represents the most mature ecosystem for the Laboratory Aptamer-Based Library Preparation Kits Market, accounting for nearly 41.8% of global consumption volume in 2025. The region benefits from a strong concentration of biotechnology firms, pharmaceutical research centers, and academic genomic laboratories. The United States alone hosts more than 1,600 active sequencing laboratories, while Canada has expanded its national genomic programs with more than 120 research facilities focusing on molecular diagnostics and drug discovery. Key industries driving demand include pharmaceutical research, oncology biomarker development, and precision medicine programs. Regulatory agencies such as the U.S. Food and Drug Administration have strengthened frameworks for genomic testing validation, encouraging laboratories to adopt standardized molecular preparation kits. Advanced technologies including robotic liquid-handling platforms and AI-assisted sequencing workflows are increasingly integrated into laboratory operations, improving experimental throughput by nearly 35%. A notable regional player, Illumina, continues to expand sequencing platform capabilities and reagent compatibility, enabling laboratories to conduct large-scale genomic experiments more efficiently. Regional consumer behavior shows higher enterprise adoption in healthcare and biotechnology research institutions, with nearly 63% of large laboratories utilizing automated nucleic acid preparation systems to improve research productivity.
Europe holds approximately 29.6% share of the Laboratory Aptamer-Based Library Preparation Kits Market, supported by strong government-backed research initiatives and cross-border biomedical collaborations. Germany, the United Kingdom, and France collectively represent more than 60% of the region’s genomic research capacity, hosting numerous biotechnology clusters and sequencing facilities. European regulatory bodies have implemented strict guidelines for molecular diagnostics and genomic testing to ensure quality and traceability in laboratory workflows. Sustainability initiatives also influence laboratory procurement strategies, with research institutions targeting 20% reductions in chemical reagent waste by 2030 through optimized molecular biology protocols. Adoption of advanced sequencing and nucleic acid enrichment technologies is growing across leading research hubs including Berlin, Cambridge, and Paris. Regional companies such as QIAGEN, headquartered in Germany, are developing integrated nucleic acid preparation technologies and automated sequencing-compatible kits to streamline genomic analysis workflows. Consumer behavior variations show that regulatory pressure and data transparency requirements drive demand for standardized laboratory kits and traceable reagent systems, encouraging laboratories across Europe to adopt highly validated aptamer-based preparation technologies.
Asia-Pacific is the fastest expanding regional ecosystem in the Laboratory Aptamer-Based Library Preparation Kits Market and ranks second in total research activity volume. The region hosts more than 3,000 biotechnology startups and research laboratories, with China, Japan, and India representing the largest consuming markets. China alone accounts for nearly 38% of the region’s genomic sequencing infrastructure, supported by significant government investment in biotechnology innovation parks and national precision medicine programs. Japan maintains strong capabilities in molecular diagnostics research, with more than 200 specialized biomedical laboratories focusing on nucleic acid technologies. India is emerging as a biotechnology research hub with over 900 biotechnology companies and research institutes engaged in genomic and molecular biology studies. Infrastructure expansion includes automated sequencing laboratories and high-throughput research facilities capable of analyzing thousands of genomic samples weekly. Regional innovation hubs such as Shenzhen, Tokyo, and Bangalore are driving technological advancements in nucleic acid screening technologies. A notable regional player, Takara Bio, continues to develop advanced molecular biology reagents and library preparation kits for genomic research laboratories. Regional consumer behavior indicates that growth is driven by expanding biotechnology startups, academic research institutions, and government-funded genome sequencing initiatives.
South America contributes approximately 6.7% of global demand for Laboratory Aptamer-Based Library Preparation Kits, with Brazil and Argentina representing the most active biotechnology research environments in the region. Brazil accounts for nearly 55% of the regional molecular biology research infrastructure, supported by national programs aimed at expanding biomedical innovation and disease surveillance systems. Argentina hosts more than 70 biotechnology research institutes conducting genomic analysis and molecular diagnostics studies. Infrastructure investments are gradually improving laboratory capabilities, with new genomic sequencing centers being established in São Paulo and Buenos Aires. Governments are introducing research funding incentives and tax benefits for biotechnology startups to encourage innovation and collaboration between academic institutions and pharmaceutical companies. Regional laboratories increasingly utilize automated nucleic acid preparation technologies to improve research productivity and experimental accuracy. Consumer behavior variations indicate that demand is closely linked to medical research institutions and public health laboratories, where molecular diagnostics and infectious disease research programs are expanding rapidly across the region.
The Middle East & Africa region accounts for approximately 4.3% of global activity in the Laboratory Aptamer-Based Library Preparation Kits Market, with increasing investments in biomedical research infrastructure across several countries. The United Arab Emirates and Saudi Arabia are leading biotechnology development initiatives, establishing advanced genomics laboratories and research centers focused on personalized medicine and disease genomics. The UAE has launched multiple national genome programs targeting the sequencing of over one million citizens, which requires extensive molecular biology workflows including nucleic acid library preparation. South Africa represents the most active biotechnology research hub in Africa, hosting more than 80 genomics and molecular biology laboratories supporting academic and clinical research programs. Technological modernization trends include integration of high-throughput sequencing platforms and automated sample preparation systems. Trade partnerships with global biotechnology companies are enabling regional research institutions to access advanced molecular biology reagents and technologies. Regional consumer behavior shows that adoption is primarily driven by government-funded research programs and academic collaborations focused on genomic medicine and disease surveillance initiatives.
United States – 36.4% Market Share: Leads due to strong biotechnology R&D infrastructure, extensive sequencing facilities, and high pharmaceutical research activity.
Germany – 11.2% Market Share: It is supported by advanced molecular diagnostics research programs and a dense network of biotechnology laboratories across Europe.
The Laboratory Aptamer-Based Library Preparation Kits Market is characterized by a moderately consolidated competitive structure with approximately 45–50 active global and regional biotechnology companies supplying molecular biology reagents, sequencing library preparation kits, and nucleic acid enrichment technologies. The top five companies collectively account for nearly 52–55% of the global market, reflecting strong brand recognition, established distribution networks, and advanced research capabilities in genomic technologies.
Leading participants focus heavily on research and development investments, particularly in automated sequencing workflows, AI-assisted biomarker discovery platforms, and microfluidic SELEX technologies. Many biotechnology firms are introducing integrated molecular biology solutions that combine library preparation kits with sequencing reagents and bioinformatics analysis tools. Product innovation remains a major competitive factor, with several companies launching automated nucleic acid preparation systems capable of processing 96 to 384 samples per experiment, significantly improving laboratory efficiency.
Strategic collaborations between biotechnology companies and academic research institutions are also shaping the competitive environment. More than 120 collaborative research programs worldwide currently utilize advanced aptamer screening platforms for drug discovery and diagnostic development. Mergers, acquisitions, and joint ventures are increasingly common as companies attempt to expand their presence in high-growth biotechnology markets such as Asia-Pacific. Additionally, companies are strengthening manufacturing capabilities and supply chain networks to support the growing global demand for sequencing reagents and nucleic acid preparation kits.
Thermo Fisher Scientific Inc.
Agilent Technologies Inc.
QIAGEN N.V.
New England Biolabs
Takara Bio Inc.
Merck KGaA (MilliporeSigma)
Integrated DNA Technologies
Bio-Rad Laboratories Inc.
Promega Corporation
TriLink BioTechnologies
Aptagen LLC
Base Pair Biotechnologies
SomaLogic Inc.
Technological innovation is playing a pivotal role in the evolution of the Laboratory Aptamer-Based Library Preparation Kits Market, particularly as genomic research laboratories demand faster, more accurate, and scalable molecular biology workflows. One of the most significant technological developments is the integration of microfluidic SELEX systems, which automate aptamer selection processes traditionally performed manually. These systems can process thousands of nucleic acid sequences simultaneously, reducing experimental cycles from approximately 12–15 rounds to fewer than 8 rounds while significantly improving candidate selection accuracy.
Automation technologies are also transforming laboratory operations. Robotic liquid-handling platforms and automated sample preparation stations are capable of processing up to 384 nucleic acid samples per run, improving throughput and minimizing manual contamination risks. These automated systems can reduce reagent consumption by nearly 35%, which supports cost-efficient laboratory operations and sustainability objectives.
Artificial intelligence is emerging as another transformative technology in the aptamer screening ecosystem. AI-powered bioinformatics platforms can analyze sequencing datasets containing millions of nucleotide sequences, enabling researchers to rapidly identify potential aptamer candidates with high binding specificity. Machine learning algorithms can improve aptamer selection efficiency by nearly 30–40% by predicting molecular interactions between nucleic acid sequences and protein targets.
Next-generation sequencing (NGS) integration is also accelerating the adoption of advanced library preparation kits. Modern sequencing instruments generate terabytes of genomic data weekly, requiring high-performance nucleic acid preparation systems capable of supporting complex genomic workflows. Several biotechnology companies are developing sequencing-compatible kits optimized for targeted enrichment and biomarker discovery.
Another emerging innovation involves nanopore sequencing technologies, which allow real-time analysis of nucleic acid molecules without requiring extensive amplification steps. Laboratories adopting nanopore sequencing can analyze genomic samples in a matter of hours rather than days, increasing demand for compatible aptamer-based library preparation technologies. These technological advancements collectively enable faster biomarker discovery, improved molecular diagnostics research, and more efficient drug development workflows across the biotechnology industry.
• In October 2025, Illumina introduced the Illumina 5-Base DNA Prep and 5-Base DNA Prep with Enrichment kits, enabling simultaneous detection of genomic variants and DNA methylation at single-base resolution. The kits support targeted enrichment workflows and are compatible with NovaSeq and NextSeq sequencing systems, improving multiomic analysis efficiency for genomic research laboratories. Source: www.illumina.com
• In September 2025, Illumina launched Illumina Protein Prep, a proteomic sample preparation workflow capable of measuring up to 9,500 unique human proteins, enabling deeper multiomics discovery and improved integration between sequencing workflows and protein analysis platforms used in biomedical research laboratories.
• In May 2024, QIAGEN launched the QIAseq Multimodal DNA/RNA Library Kit, allowing researchers to prepare DNA and RNA sequencing libraries from a single biological sample for whole-genome and transcriptome sequencing. The kit simplifies multiomics workflows used in precision medicine and cancer research programs.
• In February 2025, Illumina announced development of Constellation Mapped Reads technology, designed to streamline whole-genome sequencing workflows and improve mapping of complex genomic regions while reducing reliance on traditional library preparation processes in some sequencing applications.
The Laboratory Aptamer-Based Library Preparation Kits Market Report provides a comprehensive evaluation of the global landscape for nucleic acid library preparation technologies used in genomic research, drug discovery, and molecular diagnostics. The report examines a wide range of product types including SELEX-based library preparation kits, automated microfluidic preparation systems, and next-generation sequencing-compatible aptamer enrichment kits, each serving distinct research workflows in biotechnology laboratories.
The scope of the report covers major application areas such as biomarker discovery, pharmaceutical drug development, molecular interaction studies, and diagnostic research programs. These applications represent the primary drivers of demand for aptamer-based nucleic acid libraries, particularly within genomic laboratories that process thousands of biological samples annually.
From an end-user perspective, the report analyzes adoption patterns across academic research institutes, biotechnology companies, pharmaceutical organizations, and clinical diagnostic laboratories. Universities and government research laboratories collectively represent a substantial portion of experimental genomic research activities, while biotechnology firms and pharmaceutical companies increasingly deploy high-throughput screening technologies to accelerate therapeutic development.
Geographically, the report evaluates the market across North America, Europe, Asia-Pacific, South America, and the Middle East & Africa, examining regional biotechnology infrastructure, research funding programs, and technological innovation hubs. Countries with large genomic research programs and strong biotechnology ecosystems are analyzed in detail to highlight their influence on global demand for aptamer library preparation technologies.
Additionally, the report investigates emerging trends such as AI-assisted biomarker discovery, automated sequencing workflows, nanopore sequencing integration, and microfluidic nucleic acid screening platforms. These technological innovations are shaping the next generation of molecular biology tools used by research laboratories worldwide. The report also explores niche segments such as aptamer-based diagnostic assay development and targeted proteomics screening, offering decision-makers valuable insights into evolving research technologies and future opportunities within the Laboratory Aptamer-Based Library Preparation Kits Market.
| Report Attribute / Metric | Details |
|---|---|
| Market Revenue (2025) | USD 198.4 Million |
| Market Revenue (2033) | USD 632.7 Million |
| CAGR (2026–2033) | 15.6% |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Historic Period | 2021–2025 |
| Segments Covered |
By Type
By Application
By End-User Insights
|
| Key Report Deliverables | Revenue Forecast; Market Trends; Growth Drivers & Restraints; Technology Insights; Segmentation Analysis; Regional Insights; Competitive Landscape; Regulatory & ESG Overview; Recent Developments |
| Regions Covered | North America; Europe; Asia-Pacific; South America; Middle East & Africa |
| Key Players Analyzed | Illumina Inc.; Thermo Fisher Scientific Inc.; Agilent Technologies Inc.; QIAGEN N.V.; New England Biolabs; Takara Bio Inc.; Merck KGaA (MilliporeSigma); Integrated DNA Technologies; Bio-Rad Laboratories Inc.; Promega Corporation; TriLink BioTechnologies; Aptagen LLC; Base Pair Biotechnologies; SomaLogic Inc. |
| Customization & Pricing | Available on Request (10% Customization Free) |
