Reports
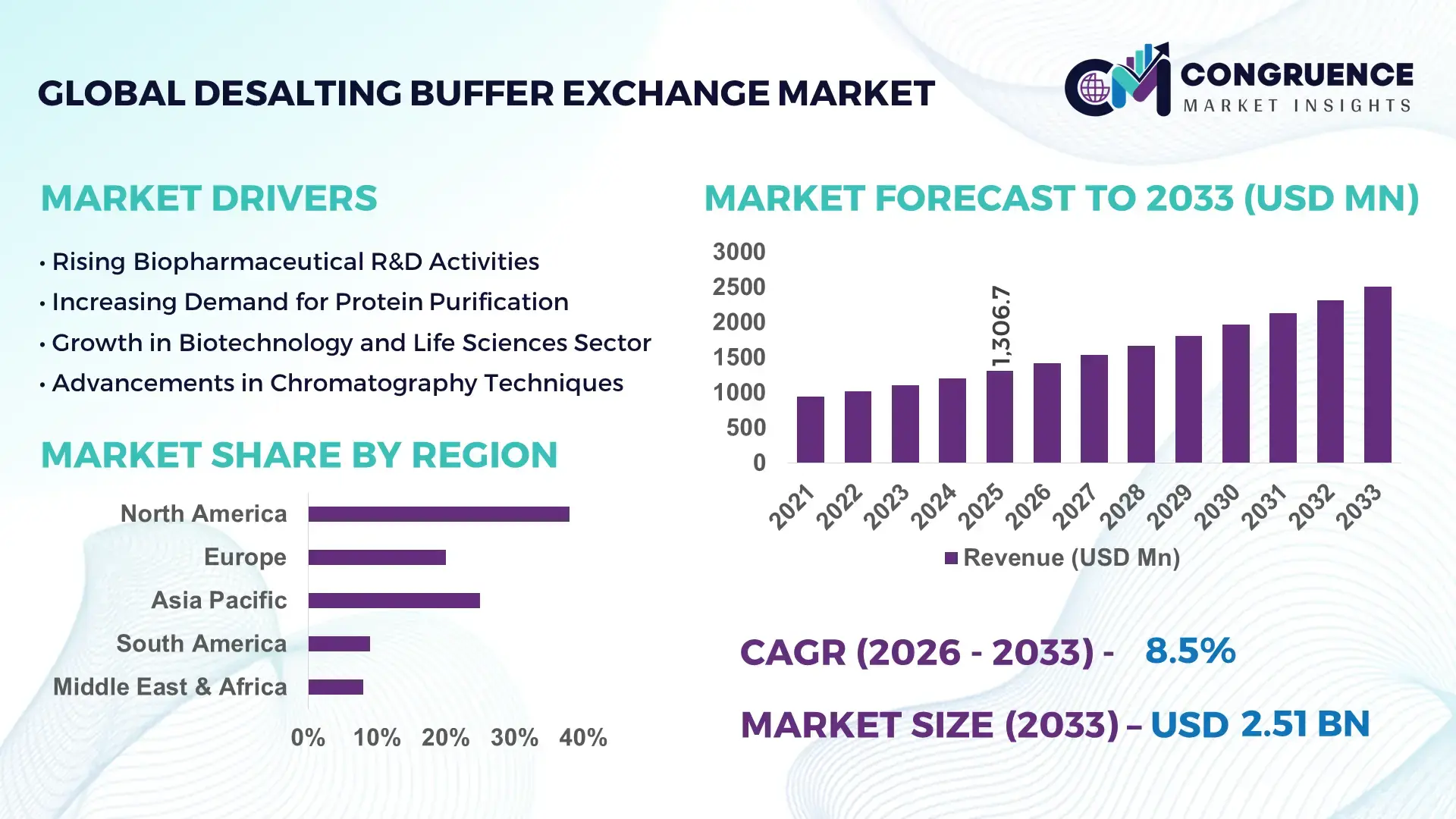
The Global Desalting Buffer Exchange Market was valued at USD 1306.71 Million in 2025 and is anticipated to reach a value of USD 2509.69 Million by 2033 expanding at a CAGR of 8.5% between 2026 and 2033. This growth is primarily driven by the increasing demand for high-purity biomolecule processing in pharmaceutical and biotechnology applications.
The United States continues to exhibit a strong position in the desalting buffer exchange market, supported by advanced biopharmaceutical manufacturing infrastructure and substantial R&D investments exceeding USD 90 billion annually in life sciences. The country operates more than 1,200 biopharma production facilities, many of which integrate automated desalting and buffer exchange systems to enhance purification efficiency. Over 65% of protein purification workflows in U.S.-based laboratories utilize advanced chromatography and membrane filtration technologies, with desalting processes embedded within them. Additionally, increasing adoption of single-use technologies has improved process turnaround times by approximately 30%, further strengthening operational capabilities across research and industrial applications.
Market Size & Growth: Valued at USD 1306.71 million in 2025, projected to reach USD 2509.69 million by 2033, growing at 8.5% CAGR due to rising demand for efficient protein purification workflows.
Top Growth Drivers: Biopharma production expansion (45%), increasing R&D activities (38%), adoption of automation technologies (32%).
Short-Term Forecast: By 2028, process efficiency in buffer exchange operations is expected to improve by 25% through automation and integrated systems.
Emerging Technologies: Single-use filtration systems, membrane chromatography innovations, and AI-driven process optimization tools.
Regional Leaders: North America projected at USD 980 million by 2033 with advanced lab infrastructure; Europe at USD 720 million with strong regulatory compliance; Asia-Pacific at USD 650 million driven by rapid biotech expansion.
Consumer/End-User Trends: Biopharmaceutical companies account for over 60% of usage, followed by academic research institutions and contract research organizations.
Pilot or Case Example: In 2024, a biotech firm improved purification efficiency by 28% through automated buffer exchange integration.
Competitive Landscape: Market leader holds approximately 22% share, followed by key players including major biotech equipment providers and filtration system manufacturers.
Regulatory & ESG Impact: Increasing adoption of eco-friendly filtration systems reducing chemical waste by up to 20%.
Investment & Funding Patterns: Over USD 2.5 billion invested globally in bioprocessing infrastructure and purification technologies.
Innovation & Future Outlook: Integration of AI-based monitoring and continuous processing systems is expected to redefine efficiency standards.
The desalting buffer exchange market is witnessing significant contributions from pharmaceutical manufacturing, biotechnology research, and clinical diagnostics, collectively accounting for over 75% of total demand. Recent advancements in membrane filtration technologies and automated chromatography systems have improved processing throughput by up to 35%, enhancing scalability for large-volume production. Regulatory frameworks emphasizing purity standards and waste reduction are accelerating the adoption of sustainable buffer exchange solutions. Regionally, Asia-Pacific is emerging as a high-growth consumption hub due to expanding biopharma manufacturing capacity and increasing government funding. Future outlook indicates strong momentum driven by continuous processing innovations and integration of smart monitoring systems.
The desalting buffer exchange market holds significant strategic relevance as a critical component in downstream bioprocessing, particularly in protein purification and biologics manufacturing. With the global biologics pipeline expanding by over 12% annually, efficient buffer exchange systems have become essential for maintaining product integrity and meeting regulatory standards. Advanced membrane filtration technology delivers nearly 40% improvement in processing speed compared to traditional dialysis methods, enabling faster turnaround and reduced operational costs.
North America dominates in volume due to its mature pharmaceutical manufacturing ecosystem, while Asia-Pacific leads in adoption with over 55% of emerging biotech enterprises integrating modern buffer exchange technologies into their workflows. This shift is supported by increased investment in biosimilar production and contract manufacturing facilities across countries such as China and India.
By 2028, AI-enabled process optimization is expected to improve purification efficiency by up to 30%, reducing manual intervention and minimizing error rates. Firms are committing to sustainability goals, targeting a 25% reduction in buffer waste and chemical usage by 2030 through adoption of reusable and single-use hybrid systems. In 2024, a leading biopharmaceutical company in Germany achieved a 22% reduction in processing time by implementing automated desalting units integrated with real-time monitoring tools. The desalting buffer exchange market is evolving as a foundational pillar supporting resilient supply chains, regulatory compliance, and sustainable manufacturing practices, positioning it as a key enabler of future-ready bioprocessing ecosystems.
The increasing demand for biologics, including monoclonal antibodies and recombinant proteins, is a major driver of the desalting buffer exchange market. Over 50% of newly approved drugs are biologics, necessitating highly efficient purification processes to ensure product stability and efficacy. Desalting and buffer exchange are essential steps in removing unwanted salts and adjusting solution conditions during downstream processing. Advanced filtration systems have demonstrated up to 35% improvement in throughput, significantly enhancing production efficiency. Additionally, the expansion of biosimilar manufacturing, particularly in emerging economies, has increased the need for cost-effective and scalable buffer exchange technologies. The growth of contract development and manufacturing organizations has further accelerated adoption, as these facilities require standardized and high-performance purification systems.
High capital investment associated with advanced desalting buffer exchange systems remains a significant restraint. Automated chromatography units and membrane filtration systems can cost between USD 50,000 and USD 500,000 depending on capacity and functionality, limiting adoption among small and mid-sized laboratories. Maintenance expenses and the need for skilled operators further increase operational costs. Additionally, integration of these systems into existing workflows often requires infrastructure upgrades, adding to the financial burden. In developing regions, limited access to funding and lack of technical expertise hinder widespread implementation. These cost-related challenges create barriers to entry, particularly for emerging biotech firms and academic research institutions operating under constrained budgets.
The adoption of single-use technologies presents significant growth opportunities in the desalting buffer exchange market. These systems reduce contamination risks by eliminating the need for cleaning and sterilization, improving operational efficiency by up to 30%. Single-use filtration units are gaining traction in small-batch and personalized medicine production, where flexibility and rapid turnaround are critical. The global shift toward modular and scalable manufacturing facilities is further supporting demand for disposable buffer exchange solutions. Additionally, innovations in membrane materials have enhanced filtration performance, enabling higher recovery rates and reduced processing times. Emerging markets are also embracing these technologies due to lower upfront infrastructure requirements, creating new avenues for market expansion.
Regulatory complexities pose a major challenge for the desalting buffer exchange market, particularly in pharmaceutical manufacturing where strict compliance standards must be met. Validation of purification processes requires extensive documentation and testing, increasing development timelines by up to 20%. Differences in regulatory frameworks across regions further complicate global operations, requiring manufacturers to adapt processes to meet varying standards. Additionally, ensuring consistency and reproducibility in buffer exchange processes is critical for product approval, necessitating advanced monitoring and control systems. Smaller companies often struggle to meet these requirements due to limited resources and expertise, which can delay product commercialization and restrict market participation.
• Rapid adoption of automated buffer exchange systems improving efficiency by over 30%
Automation is significantly transforming desalting buffer exchange workflows, with over 62% of large-scale biopharmaceutical facilities integrating automated systems into purification lines. These systems have demonstrated a reduction in manual intervention by nearly 40% and improved batch processing consistency by approximately 28%. Advanced automation platforms incorporating programmable logic controls and real-time monitoring are enabling continuous operations, reducing processing errors by 22%. North America and Europe account for more than 58% of automated system deployments, driven by the need for scalable biologics production and stringent quality control standards.
• Increasing penetration of single-use technologies with 45% adoption in bioprocessing facilities
Single-use desalting and buffer exchange technologies are gaining strong traction, with adoption rates exceeding 45% across mid- to large-scale laboratories. These systems reduce cleaning and sterilization requirements, lowering operational downtime by nearly 35%. Disposable filtration units and pre-packed columns have improved turnaround times by up to 27%, making them highly suitable for small-batch and personalized medicine applications. Asia-Pacific is witnessing rapid growth, with nearly 38% of newly established biomanufacturing facilities incorporating single-use solutions due to lower infrastructure requirements and faster deployment timelines.
• Expansion of membrane-based filtration technologies delivering 25% higher throughput
Membrane-based desalting solutions are increasingly replacing traditional dialysis methods, accounting for nearly 52% of modern buffer exchange processes. These systems provide up to 25% higher throughput and reduce processing time by approximately 30%, enhancing productivity in large-scale manufacturing. Innovations in ultrafiltration and nanofiltration membranes have improved protein recovery rates to over 90%, while minimizing sample loss. Europe leads in membrane technology innovation, with over 48% of research institutions focusing on next-generation filtration materials and performance optimization techniques.
• Integration of AI-driven monitoring tools enhancing process accuracy by 20%
The integration of artificial intelligence and data analytics into desalting buffer exchange systems is emerging as a key trend, with nearly 33% of advanced facilities deploying AI-based monitoring solutions. These tools improve process accuracy by approximately 20% and enable predictive maintenance, reducing equipment downtime by 18%. Machine learning algorithms analyze real-time process parameters to optimize buffer exchange efficiency and detect anomalies. In 2024, several pilot implementations demonstrated a 24% improvement in process reproducibility, highlighting the growing importance of digital transformation in bioprocessing environments.
The desalting buffer exchange market demonstrates a well-defined segmentation structure across product types, applications, and end-user industries, reflecting its diverse operational scope in bioprocessing and laboratory workflows. Product segmentation is primarily driven by the adoption of filtration systems, chromatography-based solutions, and dialysis techniques, each catering to varying throughput and precision requirements. Application segmentation highlights strong demand from pharmaceutical manufacturing, biotechnology research, and clinical diagnostics, collectively accounting for a majority of utilization due to their reliance on high-purity biomolecule processing. End-user segmentation reveals significant participation from biopharmaceutical companies, research institutions, and contract manufacturing organizations, with adoption patterns influenced by production scale and regulatory requirements. The increasing shift toward automated and single-use systems is reshaping segmentation dynamics, enabling faster processing cycles and improved operational flexibility across all categories.
Filtration-based systems dominate the desalting buffer exchange market, accounting for approximately 48% of total adoption due to their high efficiency, scalability, and ability to handle large sample volumes. These systems, particularly ultrafiltration and diafiltration units, are widely used in industrial-scale bioprocessing, offering protein recovery rates exceeding 90%. In comparison, chromatography-based desalting solutions hold around 32% share, valued for their precision and suitability in analytical and small-scale purification workflows. However, membrane filtration technologies are also the fastest-growing segment, expanding at an estimated 9.2% CAGR, driven by advancements in membrane materials and increased demand for high-throughput systems.
Dialysis-based desalting methods contribute the remaining 20% share, primarily used in academic and small laboratory settings due to their simplicity and cost-effectiveness, though they are less efficient for large-scale operations. Emerging hybrid systems that combine filtration and chromatography are also gaining attention, offering improved flexibility and reduced processing time.
Pharmaceutical manufacturing represents the leading application segment, accounting for approximately 54% of total usage due to the critical role of desalting buffer exchange in biologics production and drug formulation processes. Biotechnology research follows with a 28% share, driven by increasing experimentation in protein analysis, genomics, and cell biology. Clinical diagnostics holds around 18%, focusing on sample preparation and purification for diagnostic assays. Among these, biotechnology research is the fastest-growing application segment, expanding at an estimated 10.1% CAGR, supported by rising investments in life sciences research and the growing number of academic and commercial research projects.
The demand for desalting buffer exchange solutions in pharmaceutical manufacturing is further amplified by the increasing pipeline of biologics, where precise buffer conditions are essential for maintaining drug stability and efficacy. In contrast, diagnostic applications are benefiting from advancements in rapid testing technologies and increased demand for high-throughput sample processing.
Biopharmaceutical companies are the dominant end-users in the desalting buffer exchange market, accounting for approximately 57% of total adoption due to their extensive use of purification technologies in large-scale drug manufacturing. Contract research and manufacturing organizations (CROs and CMOs) follow with a 23% share, benefiting from increased outsourcing of drug development and production processes. Academic and research institutions contribute around 20%, primarily focusing on experimental and small-scale applications. Among these, CROs and CMOs represent the fastest-growing end-user segment, expanding at an estimated 9.8% CAGR, driven by the rising demand for cost-effective and scalable outsourcing solutions.
Biopharmaceutical companies are investing heavily in advanced desalting systems to enhance production efficiency and meet stringent regulatory requirements. Meanwhile, CROs and CMOs are rapidly adopting automated and single-use technologies to improve service delivery and reduce operational costs. Academic institutions continue to play a crucial role in early-stage research, contributing to innovation and technology development.
Region North America accounted for the largest market share at 38% in 2025 however, Asia-Pacific is expected to register the fastest growth, expanding at a CAGR of 9.8% between 2026 and 2033.
North America’s dominance is supported by over 1,200 active biopharmaceutical production facilities and more than 65% adoption of automated desalting buffer exchange systems. Europe holds approximately 29% share, driven by strong regulatory compliance frameworks and advanced research infrastructure across Germany, the UK, and France. Asia-Pacific contributes nearly 24% of the global volume, with China and India collectively accounting for over 55% of regional demand due to rapid expansion in biologics manufacturing. South America and the Middle East & Africa together represent around 9% share, with increasing investments in healthcare infrastructure and laboratory modernization. Globally, more than 70% of large-scale laboratories now integrate desalting buffer exchange technologies into purification workflows, reflecting rising demand for high-efficiency processing systems.
How are advanced biopharma ecosystems accelerating high-efficiency purification technologies?
North America holds approximately 38% of the desalting buffer exchange market share, supported by a mature biopharmaceutical sector and high R&D intensity. The region’s demand is primarily driven by pharmaceutical manufacturing, biotechnology research, and contract manufacturing organizations, which collectively account for over 72% of total usage. Regulatory frameworks such as stringent FDA guidelines have led to over 60% of facilities adopting validated and automated purification workflows. Digital transformation is a key trend, with nearly 48% of laboratories integrating AI-based monitoring tools to improve process accuracy and reduce operational errors. A notable example includes a U.S.-based bioprocessing equipment manufacturer that introduced automated filtration systems, improving throughput efficiency by 26% across multiple facilities. Consumer behavior reflects high enterprise adoption, particularly in healthcare and life sciences, where scalability and compliance are critical.
Why are strict compliance frameworks driving demand for precision purification systems?
Europe accounts for nearly 29% of the desalting buffer exchange market, with Germany, the UK, and France leading regional adoption. The market is heavily influenced by regulatory bodies enforcing stringent quality and environmental standards, resulting in over 58% of facilities adopting sustainable and low-waste purification technologies. Key industries include pharmaceutical manufacturing and academic research, contributing more than 68% of total demand. The region is also witnessing strong adoption of membrane filtration systems, with nearly 46% of laboratories investing in next-generation filtration technologies. A European-based life sciences company has recently implemented eco-friendly buffer exchange systems, reducing chemical waste by 21% across its operations. Consumer behavior in this region is shaped by regulatory pressure, leading to increased demand for highly accurate and compliant desalting buffer exchange solutions.
What factors are accelerating rapid expansion in high-volume bioprocessing environments?
Asia-Pacific ranks as the fastest-growing region and contributes approximately 24% of global market volume. China, India, and Japan dominate regional consumption, collectively accounting for over 65% of demand due to expanding biopharmaceutical manufacturing capacity. The region has seen a 40% increase in newly established biotech facilities over the past five years, driving demand for scalable desalting buffer exchange systems. Infrastructure development and government-backed initiatives have accelerated adoption, with nearly 52% of new facilities incorporating automated purification technologies. A leading biotech company in China has implemented advanced membrane filtration systems, achieving a 30% increase in production efficiency. Consumer behavior in Asia-Pacific reflects rapid adoption driven by cost efficiency and increasing demand for biologics, supported by strong manufacturing growth and innovation hubs.
How are emerging healthcare investments shaping purification technology adoption?
South America accounts for approximately 5% of the desalting buffer exchange market, with Brazil and Argentina as key contributors. The region is experiencing gradual growth driven by expanding healthcare infrastructure and increasing investments in pharmaceutical manufacturing. Government initiatives promoting local drug production have led to a 22% increase in laboratory modernization projects. Key industries include biotechnology research and clinical diagnostics, contributing over 60% of regional demand. Trade policies supporting equipment imports have improved access to advanced desalting technologies. A regional laboratory equipment provider has introduced cost-effective filtration systems, improving sample processing efficiency by 18% across mid-sized facilities. Consumer behavior indicates demand is closely tied to healthcare expansion and localized production capabilities, with growing interest in affordable and scalable solutions.
How is modernization of laboratory infrastructure influencing advanced purification demand?
The Middle East & Africa region represents nearly 4% of the desalting buffer exchange market, with the UAE and South Africa leading growth. Demand is primarily driven by healthcare, biotechnology, and research sectors, supported by increasing government investments in scientific infrastructure. Over 35% of newly developed laboratories in the region have integrated modern purification technologies, including desalting buffer exchange systems. Technological modernization initiatives and international trade partnerships have improved access to advanced equipment. A regional distributor has collaborated with global manufacturers to supply high-performance filtration systems, enhancing laboratory efficiency by 20%. Consumer behavior varies, with demand influenced by healthcare expansion and research development, particularly in urban centers where modernization efforts are concentrated.
United States – 34% share: Desalting Buffer Exchange market leadership driven by advanced biopharmaceutical manufacturing capacity and high adoption of automated purification technologies.
Germany – 11% share: Strong presence in the Desalting Buffer Exchange market due to robust research infrastructure and stringent regulatory frameworks supporting high-precision processing.
The desalting buffer exchange market exhibits a moderately consolidated structure, with over 45 active global and regional competitors operating across filtration, chromatography, and laboratory equipment segments. The top five companies collectively account for approximately 55% of the total market share, reflecting strong competitive concentration among leading players. Market participants are increasingly focusing on product innovation, with more than 30% of new product launches centered on single-use and automated purification technologies. Strategic partnerships and collaborations have risen by 18% over the past three years, particularly between equipment manufacturers and biopharmaceutical companies to co-develop customized solutions.
Mergers and acquisitions remain a key strategy, with at least 12 notable transactions recorded in the past two years aimed at expanding product portfolios and geographic reach. Innovation trends are heavily focused on membrane filtration advancements and AI-enabled monitoring systems, with nearly 40% of companies investing in digital transformation initiatives. Additionally, competitive differentiation is driven by process efficiency improvements, with leading players offering solutions that enhance throughput by up to 30%. The market is characterized by continuous technological evolution, where companies compete on precision, scalability, and compliance capabilities to meet the growing demands of the biopharmaceutical industry.
Thermo Fisher Scientific
Merck KGaA
Danaher Corporation
Sartorius AG
Bio-Rad Laboratories
GE HealthCare Life Sciences
Repligen Corporation
Corning Incorporated
Agilent Technologies
Lonza Group
PerkinElmer Inc.
Pall Corporation
The desalting buffer exchange market is undergoing rapid technological transformation driven by the need for higher efficiency, scalability, and precision in bioprocessing workflows. Membrane-based ultrafiltration and diafiltration technologies currently account for over 50% of desalting processes, offering protein recovery rates exceeding 90% and reducing processing time by up to 30% compared to traditional dialysis. Advances in nanofiltration membranes have improved selectivity, enabling removal of small molecules while retaining high-value biomolecules with over 95% retention efficiency.
Automation has become a central pillar, with nearly 60% of large-scale facilities deploying automated buffer exchange systems integrated with programmable logic controllers and real-time monitoring sensors. These systems reduce manual intervention by approximately 40% and enhance reproducibility by over 25%. Continuous processing technologies are also gaining traction, with over 35% of new biopharmaceutical facilities incorporating continuous desalting workflows to minimize downtime and increase throughput by nearly 28%.
Single-use technologies represent another significant innovation area, with adoption rates surpassing 45% across mid-sized and large laboratories. Disposable filtration units and pre-packed columns reduce cross-contamination risks and lower cleaning requirements, cutting operational downtime by nearly 35%. Additionally, hybrid systems combining chromatography and membrane filtration are being introduced, delivering up to 20% improvement in purification efficiency.
Digital transformation is reshaping the landscape, with approximately 33% of advanced facilities integrating AI-driven analytics and predictive maintenance tools. These technologies enable real-time optimization of buffer exchange parameters, improving process accuracy by around 20% and reducing equipment downtime by 18%. Emerging innovations such as microfluidic desalting platforms and lab-on-chip devices are also gaining attention, particularly in research environments, offering rapid processing capabilities with sample volumes reduced by up to 70%.
• In March 2025, Thermo Fisher Scientific expanded its single-use bioprocessing portfolio by introducing advanced filtration systems designed for buffer exchange applications. These systems improved protein recovery efficiency by up to 25% and reduced process turnaround time by nearly 20%. Source: www.thermofisher.com
• In September 2024, Sartorius AG launched an upgraded ultrafiltration platform with enhanced membrane durability, increasing filtration lifecycle by 30% and enabling higher throughput for large-scale biologics production. Source: www.sartorius.com
• In January 2025, Merck KGaA introduced a next-generation chromatography resin optimized for desalting and buffer exchange, achieving up to 18% higher binding efficiency and improved process consistency in downstream purification workflows. Source: www.merckgroup.com
• In June 2024, Danaher Corporation, through its life sciences segment, deployed integrated automation solutions combining buffer exchange and monitoring systems, reducing manual intervention by 35% and improving operational accuracy across multiple production sites. Source: www.danaher.com
The desalting buffer exchange market report provides a comprehensive analysis of the global landscape, covering multiple dimensions including product types, applications, technologies, and end-user industries. The report evaluates key product segments such as membrane filtration systems, chromatography-based solutions, and dialysis techniques, which collectively account for over 95% of market utilization. It further explores application areas including pharmaceutical manufacturing, biotechnology research, and clinical diagnostics, which together represent more than 75% of overall demand due to their reliance on high-purity biomolecule processing.
Geographically, the report examines five major regions, analyzing over 25 countries with detailed insights into production capabilities, infrastructure development, and adoption rates. North America and Europe together contribute more than 65% of global demand, while Asia-Pacific is highlighted for its rapid expansion in biopharmaceutical manufacturing and increasing investment in research facilities. The report also includes emerging markets in South America and the Middle East & Africa, where adoption is growing steadily with infrastructure modernization efforts.
Technological coverage includes advanced filtration systems, single-use technologies, automation platforms, and AI-driven monitoring tools, with over 40% of new installations incorporating digital integration features. The report also addresses regulatory frameworks, sustainability initiatives, and environmental considerations, including efforts to reduce chemical waste by up to 20% through eco-friendly systems. Additionally, it highlights niche segments such as microfluidic desalting and lab-scale rapid purification tools, which are gaining traction in research environments. Overall, the scope is designed to provide decision-makers with actionable insights across the entire value chain.
| Report Attribute/Metric | Report Details |
|---|---|
|
Market Revenue in 2025 |
USD V2025 Million |
|
Market Revenue in 2033 |
USD V2033 Million |
|
CAGR (2026 - 2033) |
8.5% |
|
Base Year |
2025 |
|
Forecast Period |
2026 - 2033 |
|
Historic Period |
2021 - 2025 |
|
Segments Covered |
By Types
By Application
By End-User
|
|
Key Report Deliverable |
Revenue Forecast, Growth Trends, Market Dynamics, Segmental Overview, Regional and Country-wise Analysis, Competition Landscape |
|
Region Covered |
North America, Europe, Asia-Pacific, South America, Middle East, Africa |
|
Key Players Analyzed |
Thermo Fisher Scientific, Merck KGaA, Danaher Corporation, Sartorius AG, Bio-Rad Laboratories, GE HealthCare Life Sciences, Repligen Corporation, Corning Incorporated, Agilent Technologies, Lonza Group, PerkinElmer Inc., Pall Corporation |
|
Customization & Pricing |
Available on Request (10% Customization is Free) |
