Reports
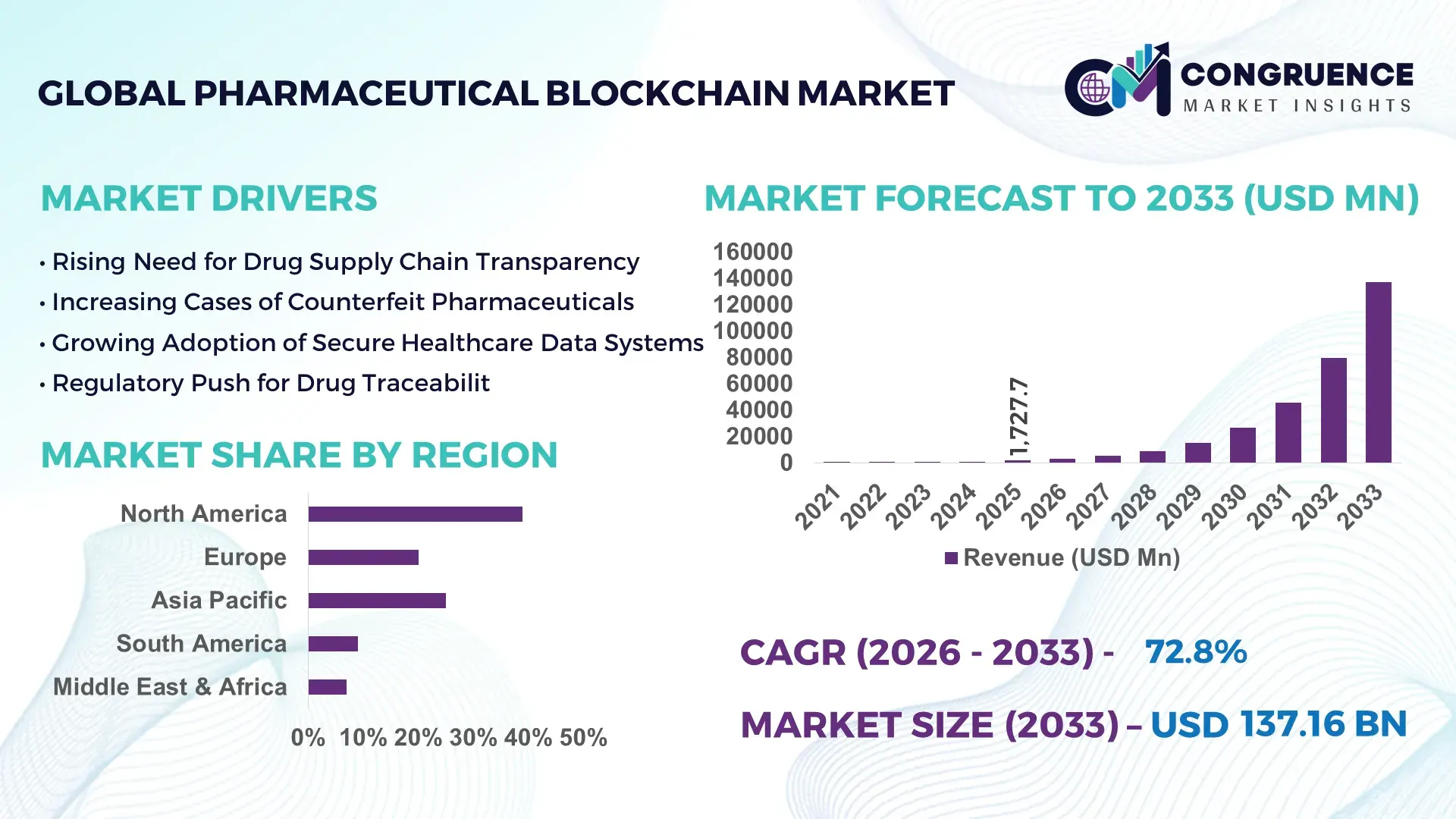
The Global Pharmaceutical Blockchain Market was valued at USD 1727.7 Million in 2025 and is anticipated to reach a value of USD 137156.05 Million by 2033 expanding at a CAGR of 72.77% between 2026 and 2033. The growth is primarily driven by rising demand for secure pharmaceutical supply chain traceability, counterfeit drug prevention, and transparent clinical data management across global healthcare systems.
The United States represents the most technologically advanced hub in the Pharmaceutical Blockchain Market, supported by strong digital health investments and a highly developed pharmaceutical ecosystem. More than 60% of large pharmaceutical manufacturers in the U.S. are actively testing blockchain-based supply chain verification systems to comply with serialization mandates under the Drug Supply Chain Security Act (DSCSA). Over USD 1.5 billion has been invested in pharmaceutical blockchain pilot initiatives since 2020 across areas such as drug traceability, clinical trial integrity, and prescription verification. The country also hosts over 40 blockchain-health startups collaborating with pharmaceutical companies to deploy decentralized data frameworks and smart contract-based supply validation platforms.
Market Size & Growth: The Pharmaceutical Blockchain Market was valued at USD 1727.7 Million in 2025 and is projected to reach USD 137156.05 Million by 2033, growing at a CAGR of 72.77%, driven by the rising need for transparent pharmaceutical supply chain authentication and anti-counterfeit drug systems.
Top Growth Drivers:
Supply chain transparency adoption increasing by 48%; pharmaceutical serialization compliance implementation reaching 55%; digital health infrastructure expansion improving operational efficiency by 37%.
Short-Term Forecast:
By 2028, blockchain-enabled drug traceability systems are expected to reduce counterfeit pharmaceutical distribution by nearly 40% and improve supply chain verification efficiency by 35%.
Emerging Technologies:
Smart contract automation, decentralized healthcare data platforms, tokenized pharmaceutical asset tracking, and AI-integrated blockchain analytics are reshaping pharmaceutical traceability and clinical data security.
Regional Leaders:
North America projected to exceed USD 60 Billion by 2033 with strong regulatory-driven adoption; Europe forecast above USD 35 Billion driven by pharmaceutical serialization initiatives; Asia-Pacific expected to surpass USD 28 Billion supported by expanding pharmaceutical manufacturing infrastructure.
Consumer/End-User Trends:
Pharmaceutical manufacturers, contract research organizations, hospital networks, and regulatory bodies increasingly adopt blockchain networks to authenticate drug origins, manage clinical trial records, and monitor cold-chain logistics.
Pilot or Case Example:
In 2024, a multinational pharmaceutical consortium deployed blockchain-based drug traceability across 12 distribution networks, achieving a 32% reduction in verification delays and a 28% improvement in supply chain visibility.
Competitive Landscape:
IBM leads the pharmaceutical blockchain platform space with approximately 24% ecosystem participation, followed by Oracle, SAP, Guardtime, and Chronicled developing distributed ledger pharmaceutical infrastructure solutions.
Regulatory & ESG Impact:
Global regulatory frameworks such as serialization mandates and digital drug tracking standards are encouraging pharmaceutical firms to adopt blockchain systems capable of reducing documentation errors by nearly 30%.
Investment & Funding Patterns:
Over USD 4.2 Billion has been invested globally in healthcare and pharmaceutical blockchain ventures between 2021 and 2025, with venture capital targeting supply chain authentication and decentralized clinical trial systems.
Innovation & Future Outlook:
Interoperable blockchain networks, AI-powered pharmaceutical compliance monitoring, and cross-border drug traceability platforms are expected to transform pharmaceutical data ecosystems and regulatory transparency worldwide.
Pharmaceutical blockchain applications are rapidly expanding across drug traceability, clinical trial data integrity, pharmaceutical logistics monitoring, and regulatory compliance automation. Supply chain traceability accounts for approximately 45% of blockchain use cases in the pharmaceutical sector, followed by clinical research data management at nearly 28%. Emerging innovations include decentralized patient consent management platforms, blockchain-secured cold chain monitoring sensors, and smart contract-driven pharmaceutical distribution networks. Growing regulatory emphasis on anti-counterfeit measures, combined with increasing cross-border pharmaceutical trade, is accelerating blockchain integration within manufacturing, wholesale distribution, and hospital procurement systems across North America, Europe, and Asia-Pacific.
The Pharmaceutical Blockchain Market has become strategically critical for pharmaceutical manufacturers and healthcare regulators seeking transparent, secure, and interoperable drug supply chains. Distributed ledger technology enables real-time drug authentication, immutable clinical trial data storage, and automated regulatory compliance reporting. Blockchain-based pharmaceutical traceability platforms deliver nearly 35% faster verification compared to traditional centralized database systems used for supply chain documentation. North America dominates pharmaceutical blockchain deployment in transaction volume due to strong pharmaceutical production and regulatory digitization, while Europe leads in adoption with nearly 46% of pharmaceutical enterprises piloting blockchain-enabled drug traceability systems across manufacturing and distribution networks.
By 2027, AI-integrated blockchain compliance monitoring is expected to reduce pharmaceutical documentation errors by nearly 30% while improving audit efficiency by approximately 25%. Companies are also committing to ESG-oriented digital transformation strategies, targeting up to 20% reduction in paper-based regulatory documentation and improved pharmaceutical waste traceability. In 2024, a pharmaceutical technology consortium in Switzerland implemented a blockchain-based supply chain platform that improved drug verification efficiency by 33% across hospital distribution networks. As digital healthcare ecosystems expand, the Pharmaceutical Blockchain Market is positioned as a cornerstone for resilient pharmaceutical logistics, regulatory transparency, and sustainable global healthcare innovation.
Counterfeit medicines remain a major challenge within global pharmaceutical distribution networks. The World Health Organization estimates that counterfeit or substandard drugs account for nearly 10% of medicines circulating in low- and middle-income countries. Blockchain-based pharmaceutical tracking platforms allow manufacturers, distributors, and pharmacies to verify product authenticity using immutable digital ledgers. Implementation of serialization mandates in over 75 countries has increased demand for blockchain-enabled traceability solutions capable of tracking millions of pharmaceutical packages across multi-tier supply chains. Smart contract technology further automates compliance verification and transaction validation, improving drug authentication efficiency while reducing manual documentation errors.
Despite strong technological potential, integrating blockchain systems into existing pharmaceutical IT infrastructure remains complex. Many pharmaceutical companies rely on legacy enterprise resource planning systems and fragmented data environments that lack interoperability with distributed ledger platforms. Studies indicate that over 55% of pharmaceutical supply chain networks still operate with partially digitized data management systems. Implementation also requires specialized blockchain expertise, cybersecurity protocols, and compliance alignment across multiple stakeholders including manufacturers, logistics providers, and regulatory authorities. These technical and operational barriers increase deployment timelines and slow adoption among small and mid-sized pharmaceutical companies.
Blockchain technology presents significant opportunities in clinical trial management by enabling secure, tamper-proof storage of patient data, consent records, and research outcomes. Approximately 70% of pharmaceutical companies report challenges related to clinical trial data transparency and auditability. Blockchain-based decentralized trial management systems can reduce data discrepancies and improve audit verification processes by maintaining immutable trial records. Integration with digital patient monitoring devices and secure patient identity verification platforms is also expanding the role of blockchain within decentralized clinical trials. As global clinical research spending exceeds USD 250 billion annually, blockchain-driven data integrity solutions are gaining attention among pharmaceutical innovators and research organizations.
Regulatory frameworks governing blockchain use in healthcare data management remain inconsistent across global jurisdictions. Pharmaceutical companies must comply with strict data privacy laws such as patient data protection regulations and cross-border data transfer rules. Blockchain’s immutable data structure can complicate compliance with “right to erasure” provisions present in many privacy regulations. Additionally, large-scale pharmaceutical blockchain networks require consensus among multiple stakeholders regarding data governance models, access permissions, and liability management. Without standardized regulatory frameworks, companies face uncertainty regarding blockchain data ownership, cross-border data sharing, and long-term compliance responsibilities within pharmaceutical supply chains.
• Pharmaceutical Supply Chain Traceability Platforms Expanding Across Global Drug Networks:
Blockchain-powered pharmaceutical traceability platforms are rapidly gaining traction as regulatory authorities push for end-to-end drug visibility. More than 65% of large pharmaceutical manufacturers have implemented digital serialization systems connected to blockchain frameworks to track product movement from production to dispensing points. These systems are capable of recording over 20 million pharmaceutical transactions per day across distributed nodes. Adoption within pharmaceutical logistics companies has increased by nearly 48% since 2022, improving real-time shipment verification and reducing counterfeit distribution risks by approximately 35%. Hospitals and wholesalers are also integrating blockchain authentication tools, enabling verification of more than 95% of serialized drug packages before reaching pharmacies.
• Smart Contract Automation Improving Pharmaceutical Compliance Operations:
Smart contract integration within blockchain platforms is transforming pharmaceutical regulatory compliance and documentation processes. Approximately 52% of pharmaceutical supply chain participants are experimenting with automated contract-based verification for drug transfers, regulatory filings, and batch release approvals. Smart contract execution has demonstrated the ability to reduce manual compliance processing time by nearly 40% while improving audit accuracy by 32%. Pharmaceutical distribution networks handling over 3 billion medicine packages annually are integrating automated verification triggers that confirm authenticity, temperature monitoring, and transport conditions. This technological shift is significantly strengthening regulatory transparency and reducing documentation errors across international pharmaceutical trade routes.
• Blockchain Integration with IoT Sensors for Cold Chain Monitoring:
Pharmaceutical companies are increasingly combining blockchain with Internet of Things (IoT) sensors to enhance cold chain monitoring for temperature-sensitive drugs and vaccines. Nearly 45% of pharmaceutical logistics providers have deployed blockchain-integrated IoT tracking devices capable of recording environmental conditions every 5 minutes during drug transportation. These platforms help ensure compliance with strict temperature requirements for biologics and vaccines, where even a 2°C deviation can compromise product safety. Implementation of real-time blockchain monitoring has reduced temperature-related product losses by approximately 28% across pharmaceutical distribution channels. More than 30 global pharmaceutical logistics networks now operate blockchain-enabled cold chain verification systems covering over 120 international shipping routes.
• Decentralized Clinical Trial Data Platforms Enhancing Research Transparency:
Blockchain adoption in clinical research is accelerating as pharmaceutical companies seek secure, transparent methods for storing trial data and patient consent records. Nearly 38% of multinational pharmaceutical firms are exploring decentralized clinical trial management platforms capable of recording millions of patient data points on immutable ledgers. Blockchain-based systems have demonstrated the potential to reduce clinical data discrepancies by nearly 29% while improving audit verification speed by 34%. Over 200 blockchain-enabled clinical research pilot programs were initiated between 2022 and 2024 across North America and Europe. These systems support encrypted patient identity management, enabling researchers to securely manage data from more than 500,000 decentralized trial participants worldwide.
The Pharmaceutical Blockchain Market is segmented based on platform type, application areas, and end-user adoption across the pharmaceutical value chain. Platform-based blockchain solutions dominate technology deployment due to their ability to support decentralized data storage and automated compliance frameworks. Application segmentation highlights strong adoption in pharmaceutical supply chain management, clinical trial data security, and drug authentication processes. Among these, supply chain traceability remains the most widely implemented use case due to increasing regulatory emphasis on counterfeit drug prevention and serialization mandates. End-user segmentation is led by pharmaceutical manufacturers and research organizations integrating blockchain systems to enhance transparency, operational efficiency, and regulatory compliance across drug production, distribution, and research operations.
The Pharmaceutical Blockchain Market by type includes public blockchain, private blockchain, and consortium blockchain platforms used to manage pharmaceutical data transactions and supply chain authentication systems. Private blockchain networks currently account for approximately 47% of total adoption across pharmaceutical enterprises due to their controlled access environments and stronger regulatory compliance capabilities. Pharmaceutical manufacturers prefer private distributed ledgers to securely manage sensitive clinical data, drug manufacturing records, and supply chain verification activities involving millions of serialized drug packages annually. Consortium blockchain platforms represent the fastest-growing segment with an estimated growth rate of about 74% annually. These networks enable collaboration among pharmaceutical companies, regulators, distributors, and healthcare providers within shared blockchain ecosystems. The ability to securely exchange pharmaceutical supply chain data across multiple stakeholders while maintaining data integrity is driving rapid adoption. Public blockchain platforms hold nearly 19% of deployments, primarily used in experimental pharmaceutical transparency initiatives and open healthcare data ecosystems. Combined, public and hybrid blockchain structures represent about 53% of the remaining infrastructure deployments across decentralized pharmaceutical research and logistics networks.
Application segmentation within the Pharmaceutical Blockchain Market includes supply chain management, clinical trials and data management, regulatory compliance, and drug authentication systems. Supply chain management dominates application usage, accounting for nearly 44% of blockchain deployments due to the growing need for real-time pharmaceutical traceability across manufacturing, distribution, and pharmacy networks. Blockchain-enabled supply chain platforms are capable of verifying millions of drug transactions daily while improving shipment visibility and preventing counterfeit medicine infiltration. Clinical trials and data management represent the fastest-growing application segment, expanding at an estimated rate of approximately 70% annually as pharmaceutical companies adopt decentralized research platforms to enhance data transparency and audit verification. Blockchain-based clinical trial systems securely store patient consent records, treatment outcomes, and research datasets while reducing data manipulation risks. Regulatory compliance automation accounts for roughly 21% of blockchain implementations, enabling automated reporting and verification for pharmaceutical serialization mandates and safety monitoring. Drug authentication and verification platforms collectively represent about 35% of additional application deployments across hospital networks and pharmacy distribution systems.
End-user segmentation in the Pharmaceutical Blockchain Market includes pharmaceutical manufacturers, contract research organizations (CROs), healthcare providers, and regulatory authorities. Pharmaceutical manufacturers represent the leading end-user segment, accounting for nearly 49% of blockchain adoption across the industry. Large pharmaceutical companies are deploying blockchain platforms to authenticate drug production batches, monitor supply chain logistics, and manage regulatory documentation associated with billions of medicine units distributed globally each year. Contract research organizations represent the fastest-growing end-user segment, expanding at an estimated rate of approximately 69% annually. CROs increasingly rely on blockchain-based clinical trial platforms to securely store research data, patient enrollment records, and trial protocols. Healthcare providers including hospitals and pharmacy networks account for approximately 28% of adoption, primarily using blockchain tools for drug verification and prescription traceability. Regulatory authorities and pharmaceutical oversight agencies collectively contribute around 23% of system adoption, integrating blockchain verification platforms to improve inspection processes and pharmaceutical import monitoring.
Region North America accounted for the largest market share at 39% in 2025 however, Asia-Pacific is expected to register the fastest growth, expanding at a CAGR of 74% between 2026 and 2033.
North America benefits from strong pharmaceutical infrastructure and advanced digital health ecosystems, with over 60% of pharmaceutical companies integrating blockchain pilots in supply chain verification. Europe holds approximately 27% share, supported by serialization regulations across 30+ countries and pharmaceutical distribution networks exceeding 8 billion medicine packages annually. Asia-Pacific accounts for nearly 22% of adoption, driven by pharmaceutical manufacturing hubs in China, India, and Japan collectively producing more than 40% of global generic medicines. South America contributes about 7% share, while the Middle East & Africa represent nearly 5%, supported by expanding digital healthcare transformation initiatives and pharmaceutical import authentication systems.
How Are Advanced Digital Healthcare Systems Accelerating Enterprise Blockchain Deployment?
North America holds approximately 39% of the Pharmaceutical Blockchain Market, driven by strong pharmaceutical manufacturing and digital healthcare infrastructure. The region hosts over 2,000 pharmaceutical manufacturing facilities, generating demand for blockchain-based drug traceability and regulatory compliance automation. The United States leads adoption due to strict drug serialization requirements under national pharmaceutical supply chain security regulations. More than 65% of pharmaceutical enterprises in the region are experimenting with blockchain platforms for drug authentication and clinical data management. Major technology firms are also advancing blockchain healthcare platforms; for example, IBM has deployed blockchain-based pharmaceutical supply chain solutions enabling verification of millions of serialized medicine packages annually. Regional consumer behavior shows higher enterprise adoption in healthcare and life sciences, with hospitals and pharmaceutical distributors increasingly relying on secure digital supply chain verification systems.
Why Are Regulatory-Driven Pharmaceutical Compliance Systems Accelerating Digital Ledger Adoption?
Europe represents nearly 27% of the Pharmaceutical Blockchain Market, supported by strict pharmaceutical traceability regulations and digital healthcare initiatives across the European Union. Countries such as Germany, the United Kingdom, and France collectively account for over 60% of regional blockchain pilot projects within pharmaceutical distribution networks. Implementation of the European pharmaceutical serialization framework has enabled tracking of more than 10 billion medicine packages annually. Pharmaceutical companies across Europe are integrating distributed ledger platforms with advanced supply chain analytics and digital product authentication technologies. A notable example includes Guardtime, a blockchain technology company operating healthcare data verification platforms supporting pharmaceutical integrity monitoring across several European healthcare networks. Consumer behavior in the region is strongly influenced by regulatory compliance requirements, leading to increased demand for transparent and verifiable pharmaceutical data management systems.
How Are Expanding Pharmaceutical Manufacturing Hubs Driving Distributed Ledger Adoption?
Asia-Pacific ranks as the second-largest and fastest-expanding region in the Pharmaceutical Blockchain Market, representing approximately 22% of global deployment activity. Major pharmaceutical manufacturing economies including China, India, and Japan collectively produce more than 40% of the world’s generic medicines, creating demand for advanced drug traceability systems. Pharmaceutical exports from the region exceed US$300 billion annually, requiring secure digital authentication of cross-border drug shipments. Technology innovation hubs in Singapore, South Korea, and India are developing blockchain-based healthcare platforms supporting pharmaceutical supply chain monitoring and clinical trial transparency. For example, Tech Mahindra has developed blockchain-powered pharmaceutical supply chain platforms used to verify drug authenticity across multi-country distribution networks. Consumer behavior in the region shows strong growth driven by digital healthcare expansion, mobile technology adoption, and increasing pharmaceutical export verification requirements.
What Role Do Pharmaceutical Distribution Networks Play in Digital Traceability Adoption?
South America represents nearly 7% of the Pharmaceutical Blockchain Market, with growing interest in digital drug authentication and pharmaceutical distribution monitoring. Brazil and Argentina account for more than 65% of pharmaceutical consumption within the region, driving adoption of blockchain-based supply chain verification tools. Pharmaceutical imports across South America exceed 1.8 billion medicine units annually, creating demand for secure tracking systems capable of preventing counterfeit drug circulation. Governments are implementing digital healthcare modernization programs to improve pharmaceutical monitoring infrastructure. Regional technology companies are collaborating with pharmaceutical distributors to develop blockchain-based verification systems capable of validating millions of serialized drug packages. Consumer behavior shows increasing demand for transparent medicine verification systems, particularly in large urban healthcare networks where pharmaceutical supply chain security has become a priority.
How Are Healthcare Digitalization Programs Encouraging Secure Pharmaceutical Data Platforms?
The Middle East & Africa Pharmaceutical Blockchain Market accounts for roughly 5% of global adoption, supported by expanding healthcare digitalization programs and pharmaceutical import monitoring initiatives. Countries including the United Arab Emirates and South Africa are leading regional blockchain healthcare innovation with smart health infrastructure investments exceeding US$10 billion across digital healthcare projects. Pharmaceutical imports represent more than 70% of total medicine consumption in several Gulf countries, increasing the need for blockchain-based drug authentication systems. Government-supported digital transformation initiatives are integrating blockchain into pharmaceutical supply chain verification and healthcare data security platforms. Regional technology firms are collaborating with pharmaceutical regulators to deploy distributed ledger solutions capable of validating millions of pharmaceutical transactions annually. Consumer behavior trends show increasing reliance on digital healthcare verification systems and secure pharmaceutical tracking technologies.
United States – 34% share in the Pharmaceutical Blockchain Market
Strong pharmaceutical manufacturing ecosystem, strict drug traceability regulations, and widespread enterprise adoption of blockchain healthcare platforms.
China – 18% share in the Pharmaceutical Blockchain Market
Large pharmaceutical production capacity, rapid healthcare digitalization, and growing government support for blockchain-enabled drug supply chain monitoring.
The Pharmaceutical Blockchain Market features a moderately fragmented competitive landscape with more than 120 active technology providers, pharmaceutical IT vendors, and blockchain solution developers participating in global deployments. The top five companies collectively account for nearly 46% of industry deployments, reflecting a mix of established enterprise technology firms and emerging blockchain innovators. Market leaders focus on developing scalable distributed ledger platforms capable of processing millions of pharmaceutical supply chain transactions daily while ensuring regulatory compliance and data security.
Strategic collaborations between technology providers and pharmaceutical manufacturers are shaping competitive positioning. Over 75 strategic partnerships have been formed since 2021 to develop blockchain-enabled drug traceability and clinical data management platforms. Product innovation is another key competitive factor, with companies introducing AI-integrated blockchain analytics tools capable of improving pharmaceutical supply chain verification efficiency by 30–35%.
Mergers, acquisitions, and consortium-based platforms are also influencing competition. Several pharmaceutical technology consortia now connect over 150 pharmaceutical manufacturers, distributors, and healthcare providers on shared blockchain networks designed to authenticate drug shipments and track serialized medicine packages. Companies investing heavily in interoperability solutions, smart contract automation, and decentralized healthcare data platforms are strengthening their market positioning as demand for secure pharmaceutical digital infrastructure continues to grow globally.
IBM
Oracle Corporation
SAP SE
Guardtime
Chronicled
Microsoft Corporation
BurstIQ
FarmaTrust
Modum
Factom
Blockpharma
VeChain
Patientory
Embleema
MediLedger
Technological innovation within the Pharmaceutical Blockchain Market is increasingly centered on distributed ledger interoperability, smart contract automation, and integration with advanced digital health technologies. Modern blockchain frameworks used in pharmaceutical supply chains are capable of processing over 50,000 secure transactions per second, enabling real-time verification of drug shipments across multi-tier distribution networks. Smart contract technology is improving pharmaceutical compliance workflows by automating batch verification, regulatory reporting, and supply chain authentication processes, reducing manual verification steps by nearly 35%.
Integration with Internet of Things (IoT) sensors is another major technological advancement. Blockchain-enabled cold-chain monitoring systems now track temperature and logistics data for millions of pharmaceutical shipments annually, ensuring compliance with strict storage requirements for biologics and vaccines. Edge-computing IoT devices connected to blockchain networks can record environmental data every 3–5 minutes, improving supply chain transparency and reducing temperature-related product losses by nearly 25%.
Artificial intelligence is also being integrated with blockchain analytics platforms to detect anomalies in pharmaceutical distribution patterns. AI-driven blockchain systems can analyze billions of serialized drug records to identify counterfeit risks or supply chain irregularities. Additionally, interoperable blockchain protocols are being developed to allow pharmaceutical companies, regulators, and logistics providers to securely exchange verified data across decentralized healthcare ecosystems, strengthening traceability and global regulatory compliance.
• In November 2024, Chronicled expanded the MediLedger Network to support full-scale compliance with the U.S. Drug Supply Chain Security Act (DSCSA). The platform enables secure verification of serialized drug identifiers across manufacturers, wholesalers, and dispensers, processing millions of product verification requests across pharmaceutical distribution systems.
Source: www.chronicled.com
• In April 2025, VeChain upgraded its ToolChain™ blockchain platform to strengthen pharmaceutical traceability capabilities, enabling companies to track temperature-sensitive drug shipments using IoT-integrated blockchain records. The upgrade enhanced authentication and transparency features used in healthcare and pharmaceutical logistics.
Source: www.vechain.org
• In June 2024, Embleema expanded its PatientTruth blockchain platform to support decentralized clinical trial data verification, allowing pharmaceutical companies to securely record patient consent and treatment outcomes using distributed ledger technology across research organizations.
Source: www.embleema.com
• In February 2025, IBM announced new enterprise enhancements to its IBM Blockchain Transparent Supply platform, enabling pharmaceutical companies to integrate AI-powered analytics with blockchain-based supply chain verification systems to track medicine provenance and improve regulatory compliance across global distribution networks.
Source: www.ibm.com
The Pharmaceutical Blockchain Market Report provides a comprehensive assessment of blockchain technology adoption across pharmaceutical manufacturing, supply chain management, clinical research, and regulatory compliance applications. The report analyzes more than 10 major technology segments, including private blockchain networks, consortium platforms, smart contract systems, decentralized clinical data platforms, and IoT-enabled pharmaceutical logistics monitoring solutions.
Coverage includes detailed segmentation across platform types, application areas, and end-user industries, evaluating the role of blockchain in drug authentication, supply chain verification, clinical trial data management, prescription validation, and pharmaceutical cold-chain monitoring. The report examines implementation trends across five key geographic regions and over 20 major pharmaceutical markets, highlighting regional digital healthcare infrastructure, regulatory frameworks, and technology adoption levels.
Industry analysis also evaluates blockchain integration with emerging technologies such as artificial intelligence analytics, IoT sensor networks, and decentralized healthcare data platforms capable of processing millions of pharmaceutical transactions daily. The study further explores evolving industry ecosystems involving pharmaceutical manufacturers, logistics providers, regulatory authorities, technology vendors, and healthcare organizations participating in blockchain-enabled drug traceability networks. The report therefore provides strategic insights for decision-makers seeking to understand the expanding role of blockchain within secure, transparent, and digitally integrated pharmaceutical supply chains.
| Report Attribute/Metric | Report Details |
|---|---|
|
Market Revenue in 2025 |
USD V2025 Million |
|
Market Revenue in 2033 |
USD V2033 Million |
|
CAGR (2026 - 2033) |
72.77% |
|
Base Year |
2025 |
|
Forecast Period |
2026 - 2033 |
|
Historic Period |
2021 - 2025 |
|
Segments Covered |
By Types
By Application
By End-User
|
|
Key Report Deliverable |
Revenue Forecast, Growth Trends, Market Dynamics, Segmental Overview, Regional and Country-wise Analysis, Competition Landscape |
|
Region Covered |
North America, Europe, Asia-Pacific, South America, Middle East, Africa |
|
Key Players Analyzed |
IBM, Oracle Corporation, SAP SE, Guardtime, Chronicled, Microsoft Corporation, BurstIQ, FarmaTrust, Modum, Factom, Blockpharma, VeChain, Patientory, Embleema, MediLedger |
|
Customization & Pricing |
Available on Request (10% Customization is Free) |
