Reports
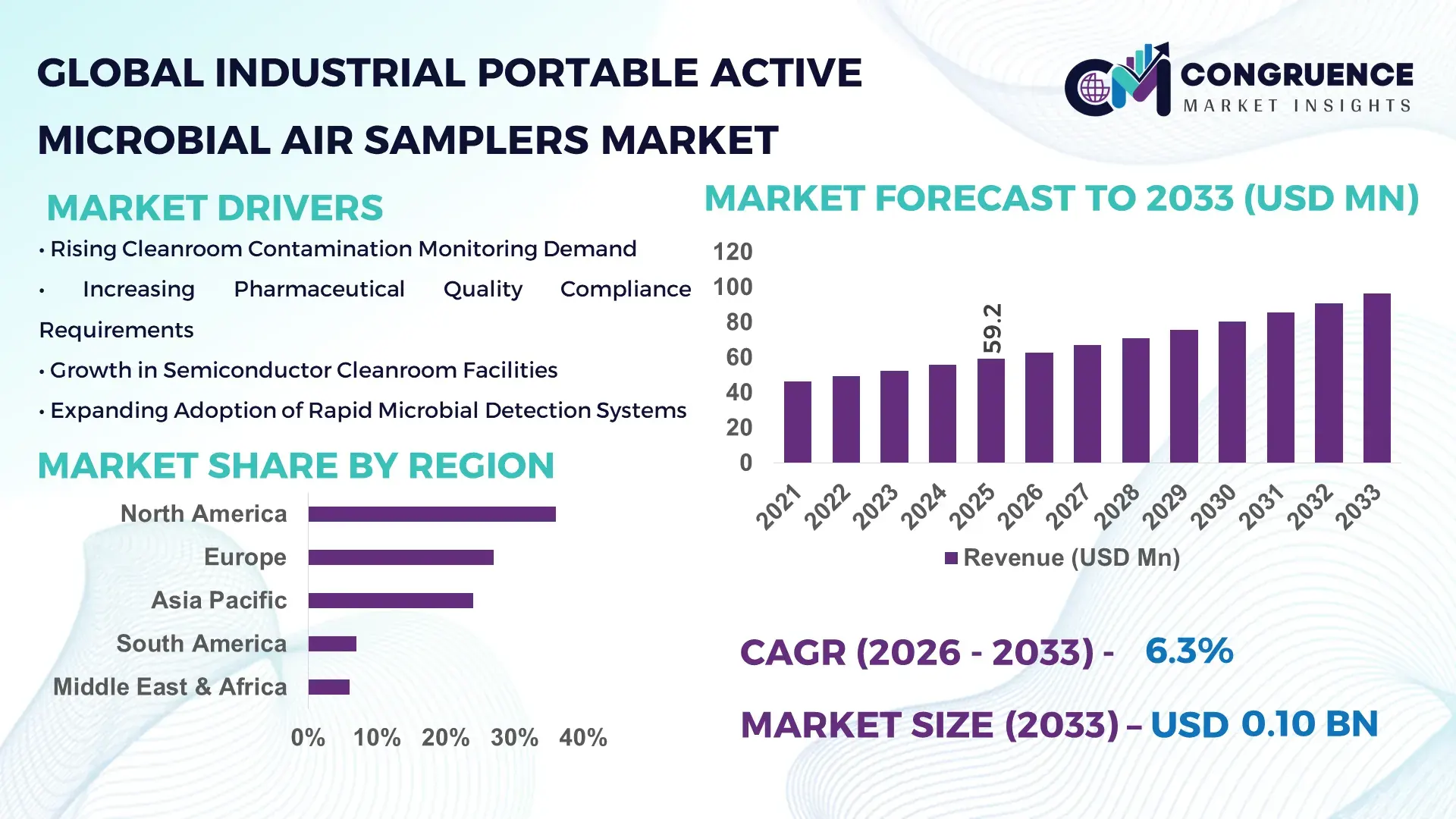
The Global Industrial Portable Active Microbial Air Samplers Market was valued at USD 59.2 Million in 2025 and is anticipated to reach a value of USD 96.5 Million by 2033 expanding at a CAGR of 6.3% between 2026 and 2033.
Rising enforcement of cleanroom contamination protocols across pharmaceutical manufacturing, semiconductor fabrication, and sterile food processing is accelerating deployment of high-efficiency portable microbial monitoring systems, particularly models integrated with rapid digital colony-count analytics and HEPA-compatible airflow calibration. Between 2024 and 2026, global biosafety regulations tightened following pharmaceutical supply chain disruptions and cross-border contamination incidents, forcing manufacturers to upgrade environmental monitoring systems with real-time traceability and portable validation capability.
The United States dominates the Industrial Portable Active Microbial Air Samplers Market with nearly 34% global demand concentration, supported by over 2,800 FDA-regulated sterile manufacturing facilities and large-scale biologics production investments exceeding USD 12 billion between 2024 and 2026. Pharmaceutical and biotechnology sectors contribute over 48% of industrial deployment volume, while semiconductor cleanroom monitoring adoption increased by 21% due to advanced chip manufacturing expansion. Compared to conventional settle-plate monitoring, portable active microbial air samplers improve airborne contamination detection sensitivity by nearly 40% while reducing sampling turnaround time by 30%. Europe remains strong in compliance-driven adoption, whereas Asia-Pacific is rapidly scaling localized manufacturing capacity and lower-cost automated monitoring systems.
Strategic competition is now shifting toward portable systems with automated data logging, rapid microbial detection, and audit-ready compliance integration, forcing suppliers to prioritize technology differentiation and faster validation cycles.
Market Size & Growth: USD 59.2 million market in 2025 reaches USD 96.5 million by 2033, driven by rapid sterile manufacturing expansion and automated contamination monitoring adoption.
Top Growth Drivers: Pharmaceutical cleanroom demand surged 28%, biologics manufacturing expanded 24%, and semiconductor contamination-control investments increased 19% globally.
Short-Term Forecast: By 2028, digital microbial monitoring workflows reduce environmental testing turnaround time by 32% and manual compliance costs by 18%.
Emerging Technologies: AI-enabled colony analytics, IoT-connected portable samplers, and rapid ATP-based detection systems improve sampling accuracy by nearly 35%.
Regional Leaders: North America exceeds USD 28 million demand, Europe strengthens GMP-driven adoption, while Asia-Pacific records 26% faster localized manufacturing deployment.
Consumer/End-User Trends: Over 57% of pharmaceutical manufacturers now prioritize portable microbial samplers with cloud-based audit traceability and automated calibration features.
Pilot/Case Example: In 2025, a biologics facility modernization project improved contamination detection efficiency by 37% through portable active air monitoring integration.
Competitive Landscape: Top five manufacturers control nearly 54% market share, led by Sartorius, Merck, bioMérieux, Particle Measuring Systems, and VWR.
Regulatory & ESG Impact: Enhanced Annex 1 sterile manufacturing compliance reduced contamination-related batch rejection rates by approximately 22% across advanced facilities.
Investment & Funding: More than USD 450 million flowed into cleanroom automation, sterile monitoring expansion, and contamination-control partnerships during 2024–2026.
Innovation & Future Outlook: Compact wireless samplers, predictive contamination analytics, and robotic cleanroom integration are redefining high-growth industrial biosafety operations.
Pharmaceutical manufacturing contributes nearly 48% of total industrial portable active microbial air sampler deployment, followed by biotechnology and semiconductor cleanrooms with a combined 34% share. Recent innovations include AI-assisted colony recognition systems improving detection precision by 31% and portable IoT-enabled samplers reducing audit documentation time by 26%. North America leads high-value compliance adoption, while Asia-Pacific is rapidly expanding localized manufacturing and sterile processing infrastructure amid global supply chain diversification. Increasing Annex 1 enforcement and accelerated biologics production are pushing companies toward automated, portable contamination-monitoring ecosystems, creating a strong foundation for strategic expansion and competitive repositioning.
Industrial portable active microbial air samplers are rapidly becoming mission-critical infrastructure across pharmaceutical manufacturing, semiconductor fabrication, biologics processing, hospital sterilization, and advanced food production environments where contamination control directly impacts operational continuity, compliance exposure, and product integrity. Competitive intensity is accelerating as manufacturers prioritize real-time environmental monitoring systems capable of reducing contamination risks, minimizing batch rejection, and strengthening audit-readiness across global production networks. The market is transforming from a compliance-driven procurement category into a strategic operational technology segment tied directly to production reliability and quality assurance performance.
Stricter sterile manufacturing regulations, including revised Annex 1 implementation and heightened FDA environmental monitoring expectations, are forcing companies to modernize legacy contamination-control workflows. AI-enabled microbial detection platforms improve operational efficiency by 38% while reducing manual analysis costs by 24% compared to traditional culture-based monitoring systems. North America leads in deployment volume and regulated pharmaceutical demand, while Asia-Pacific leads in manufacturing expansion and adoption acceleration with nearly 29% growth in localized cleanroom infrastructure investments. Over the next three years, automated portable monitoring deployment across biologics and advanced therapy manufacturing facilities is expected to improve environmental testing productivity by over 33% while reducing contamination investigation timelines by approximately 27%.
Sustainability and ESG alignment are also becoming competitive differentiators. Facilities deploying energy-efficient portable microbial samplers with digital reporting workflows reduce paper-based compliance documentation by 40% and lower repeat sampling waste through optimized monitoring cycles. In 2025, a large sterile injectables facility integrated wireless active air sampling systems across multiple cleanrooms and achieved a 35% improvement in contamination response time alongside a 19% reduction in operational deviations. Simultaneously, major suppliers are shifting capital allocation toward rapid-detection technologies, cloud-integrated monitoring ecosystems, and regional manufacturing expansion to secure long-term supply resilience. The competitive pathway ahead will be defined by who can deliver faster contamination intelligence, seamless compliance integration, and scalable portable monitoring systems capable of optimizing operational uptime while strengthening global regulatory positioning.
The Industrial Portable Active Microbial Air Samplers Market is being reshaped by rising contamination-control standards across pharmaceutical manufacturing, biologics production, semiconductor fabrication, and high-purity industrial environments. Environmental monitoring has shifted from periodic compliance testing toward continuous operational risk management, forcing manufacturers to deploy portable systems capable of delivering faster microbial detection, automated data logging, and audit-ready traceability. Demand concentration remains strongest in sterile pharmaceutical processing, which accounts for nearly half of total deployment volume, while biotechnology and semiconductor cleanrooms are accelerating adoption due to stricter particulate and airborne contamination controls. Global manufacturing decentralization and regional supply chain restructuring between 2024 and 2026 have intensified investment in localized cleanroom infrastructure, increasing demand for flexible portable monitoring systems. Companies are responding through automation integration, AI-enabled analytics, and compact wireless designs that improve monitoring speed and reduce manual intervention. Competition is increasingly defined by technology precision, compliance adaptability, and operational efficiency rather than equipment pricing alone, redefining procurement priorities across advanced industrial facilities.
The strongest growth driver in the Industrial Portable Active Microbial Air Samplers Market is the rapid expansion of sterile manufacturing environments combined with aggressive enforcement of contamination-control standards. Pharmaceutical and biotechnology facilities now account for approximately 48% of active deployment demand as global injectable drug production and advanced therapy manufacturing continue expanding. Revised Annex 1 implementation and intensified FDA environmental monitoring scrutiny increased cleanroom validation activity by nearly 27% between 2024 and 2026. Semiconductor fabrication facilities also accelerated adoption, with airborne microbial monitoring integration rising 19% as advanced chip manufacturing shifted toward ultra-clean production environments. These pressures are forcing companies to transition from passive settle-plate monitoring toward portable active systems capable of improving airborne contamination detection sensitivity by over 40%. In response, manufacturers are accelerating product innovation, increasing localized production capacity, and forming strategic partnerships with cleanroom automation providers. The direct result is faster deployment cycles, expanded recurring calibration services, and stronger integration between portable monitoring systems and digital compliance infrastructure.
Despite strong demand momentum, high validation costs and operational integration complexity continue constraining broader adoption across mid-sized industrial facilities. Advanced portable microbial air samplers equipped with automated analytics and wireless traceability systems can increase upfront deployment expenses by 22%–30% compared to conventional manual monitoring approaches. Regulatory calibration requirements and multi-site validation processes extend deployment timelines by nearly 18%, particularly in highly regulated pharmaceutical and semiconductor environments. Supply chain concentration for precision airflow sensors and sterile sampling components also remains a structural challenge, with over 60% of specialized component sourcing concentrated within a limited group of global suppliers. These pressures directly impact procurement speed, maintenance costs, and scalability across emerging industrial markets. Companies are mitigating risk through supplier diversification, modular system architectures, and long-term service agreements designed to stabilize operational costs. Some manufacturers are also introducing hybrid monitoring systems combining portable and fixed-site configurations to improve affordability without compromising compliance readiness.
The largest strategic opportunity lies in rapid microbial detection technologies integrated with smart cleanroom ecosystems. AI-driven colony recognition and ATP-based rapid testing systems are reducing environmental monitoring turnaround times by nearly 35% while improving contamination response efficiency by over 28%. Demand for IoT-connected portable samplers increased 24% during 2025 as pharmaceutical manufacturers prioritized centralized compliance dashboards and predictive contamination analytics. Emerging biologics manufacturing hubs across Asia-Pacific and the Middle East are also creating new demand pockets for compact portable systems capable of flexible deployment across modular cleanroom environments. A significant non-obvious advantage is operational labor optimization, as automated reporting workflows reduce manual compliance documentation workloads by approximately 31%. Companies are aggressively positioning through R&D investment, software integration partnerships, and ecosystem-based monitoring platforms combining microbial detection, environmental analytics, and cloud-based audit management. The competitive focus is shifting toward scalable intelligent monitoring infrastructure rather than standalone equipment sales, creating long-term recurring service and software monetization opportunities.
The market faces growing execution challenges tied to scalability, standardization, and high-performance consistency across increasingly complex industrial environments. Portable microbial air samplers deployed in multi-zone cleanroom operations often face calibration drift and airflow variability issues, impacting measurement consistency by nearly 14% in high-frequency monitoring conditions. Workforce shortages in contamination-control validation and microbiological compliance management further constrain deployment efficiency, particularly across rapidly expanding biologics facilities. Rising global cleanroom construction activity has also intensified pressure on precision component supply chains, increasing lead times for advanced portable systems by approximately 16% between 2024 and 2025. These limitations directly affect operational continuity, customer confidence, and long-term adoption scalability. Companies must solve interoperability gaps between legacy environmental monitoring systems and new digital compliance platforms while simultaneously investing in automated calibration, AI-assisted diagnostics, and regional servicing networks. Strategic partnerships with software providers, cleanroom integrators, and validation specialists are becoming essential to maintaining competitive positioning in a market increasingly defined by reliability, integration speed, and operational resilience.
31% Increase in AI-Based Contamination Analytics Adoption Reshaping Cleanroom Monitoring: Pharmaceutical and biologics manufacturers are rapidly integrating AI-enabled colony recognition systems into portable microbial air samplers to reduce manual analysis dependency and improve detection consistency. Automated image-based analytics lowered environmental monitoring review times by 29% while improving traceability accuracy by nearly 34%. Companies are expanding software partnerships and embedding cloud-based compliance dashboards to optimize audit readiness amid tightening sterile manufacturing regulations.
26% Expansion in Wireless Portable Sampling Deployment Transforming Operational Flexibility: Industrial facilities are replacing tethered microbial monitoring systems with compact wireless portable units capable of synchronized multi-room validation and remote data transfer. Deployment rates across modular cleanrooms increased 26% during 2025 as manufacturers prioritized operational agility and faster contamination investigations. The shift is reducing technician movement across controlled environments while improving sampling efficiency by approximately 21%, particularly in biologics and semiconductor production sites.
22% Growth in Localized Manufacturing Capacity Redefining Asia-Pacific Supply Dynamics: Global suppliers are accelerating regional assembly and calibration operations across Asia-Pacific to reduce delivery delays and improve cleanroom equipment responsiveness. Localized manufacturing expansion reduced lead times by nearly 18% while strengthening component availability during ongoing global supply chain restructuring. Companies are increasingly prioritizing region-specific compliance customization and lower-cost production strategies to capture rapidly scaling pharmaceutical infrastructure demand.
37% Rise in Integrated Environmental Monitoring Platforms Shifting Procurement Priorities: Industrial buyers are moving away from standalone microbial samplers toward integrated monitoring ecosystems combining airborne contamination analysis, particulate tracking, and digital compliance management. Integrated deployment improved environmental deviation response time by 37% while reducing manual reporting workloads by nearly 25%. This shift is forcing traditional equipment suppliers to expand into software-enabled service models and long-term compliance partnerships rather than competing solely on hardware specifications.
The Industrial Portable Active Microbial Air Samplers Market is segmented by type, application, and end-user, with demand increasingly concentrated around high-compliance sterile manufacturing environments and advanced contamination-control operations. Portable slit-to-agar and impaction-based systems dominate deployment due to operational reliability and compatibility with pharmaceutical cleanroom validation standards, while rapid-detection portable samplers are gaining traction as facilities prioritize faster environmental monitoring cycles. Pharmaceutical manufacturing remains the largest application segment, contributing over 48% of total deployment demand, followed by biotechnology and semiconductor cleanrooms. Hospitals, food processing plants, and research laboratories continue expanding adoption as contamination-control requirements intensify globally. Demand is shifting toward digitally integrated portable systems capable of supporting automated compliance documentation and real-time contamination analytics. Approximately 57% of industrial buyers now prioritize systems with cloud-enabled reporting and automated calibration functionality. Companies are strategically expanding product portfolios, regional servicing capabilities, and software integration partnerships to capture long-term growth across both established and emerging industrial contamination-monitoring ecosystems.
Portable impaction-based microbial air samplers dominate the Industrial Portable Active Microbial Air Samplers Market with approximately 46% share due to their strong accuracy, regulatory compatibility, and widespread integration across pharmaceutical and sterile manufacturing cleanrooms. Their structural advantage lies in high-efficiency airborne microorganism capture capability combined with validated compliance performance, making them the preferred choice for GMP-regulated facilities. Portable slit-to-agar samplers remain strategically relevant with nearly 28% share because of their continuous monitoring capability and suitability for critical aseptic processing environments. However, rapid microbial detection portable samplers represent the fastest-growing category, recording nearly 24% adoption acceleration as industrial operators prioritize shorter contamination response cycles and digital monitoring integration. Compared to traditional culture-based systems, advanced rapid-detection technologies improve contamination analysis speed by over 35% while reducing manual review dependency significantly. Remaining portable centrifugal and filtration-based systems collectively account for around 26% share, serving niche high-volume air monitoring and specialized industrial validation applications. Companies are increasingly reallocating R&D investment toward compact wireless systems, AI-assisted analytics, and automated calibration technologies, signaling a decisive shift toward intelligent portable monitoring platforms capable of optimizing operational compliance and efficiency.
Pharmaceutical manufacturing leads the Industrial Portable Active Microbial Air Samplers Market with nearly 48% application share due to stringent sterile production requirements, aggressive environmental monitoring mandates, and expanding biologics manufacturing capacity. Usage concentration remains highest in injectable drug production and aseptic processing where contamination events directly impact batch integrity and regulatory compliance. Biotechnology applications represent the fastest-growing segment with approximately 25% adoption expansion, driven by rapid cell and gene therapy production growth alongside intensified cleanroom validation requirements. Compared to mature pharmaceutical deployment, biotechnology facilities are adopting more flexible portable monitoring systems optimized for modular production environments and digital compliance integration. Semiconductor cleanrooms, food processing, hospitals, and research laboratories collectively contribute around 39% of market demand, with semiconductor manufacturing showing particularly strong adoption growth due to ultra-clean fabrication requirements. Industrial operators are increasingly shifting toward integrated monitoring workflows combining microbial sampling, particulate tracking, and automated reporting systems. Companies are responding by scaling software-enabled platforms, strengthening calibration service networks, and customizing portable monitoring solutions based on sector-specific contamination risks and operational intensity.
Pharmaceutical and biotechnology manufacturers represent the largest end-user group in the Industrial Portable Active Microbial Air Samplers Market, accounting for nearly 52% of total demand due to high-frequency environmental monitoring requirements and strict contamination-control dependency. Large-scale sterile production facilities deploy portable microbial monitoring systems extensively across cleanrooms, validation zones, and quality assurance workflows to reduce compliance exposure and operational downtime. Semiconductor manufacturers are emerging as the fastest-growing end-user category, recording approximately 23% deployment growth as advanced chip fabrication facilities intensify airborne contamination prevention investments. Compared to established pharmaceutical demand driven by compliance intensity, semiconductor adoption is fueled by process sensitivity, yield protection, and ultra-clean manufacturing precision. Hospitals, food processing facilities, and academic research laboratories collectively contribute around 34% share, increasingly prioritizing compact and digitally integrated monitoring systems. Industrial buyers are favoring long-term service contracts, predictive maintenance support, and cloud-enabled reporting functionality over standalone hardware procurement. Companies are strategically targeting high-growth end-users through customized validation solutions, scalable software ecosystems, and regional service expansion to strengthen retention and capture recurring operational revenue streams.
North America accounted for the largest market share at 36% in 2025 however, Asia-Pacific is expected to register the fastest growth, expanding at a CAGR of 7.4% between 2026 and 2033.

North America leads in regulated pharmaceutical manufacturing demand, biologics production, and advanced cleanroom compliance deployment, while Europe maintains strong adoption through strict Annex 1 environmental monitoring enforcement and sustainability-focused sterile manufacturing modernization. Asia-Pacific accounts for nearly 24% of global demand and is rapidly strengthening its position through localized production expansion, semiconductor fabrication growth, and rising pharmaceutical infrastructure investments across China, India, South Korea, and Singapore. South America and the Middle East & Africa collectively contribute over 13% share, supported by healthcare infrastructure upgrades and industrial contamination-control investments. Global supply chain diversification and regional manufacturing decentralization are accelerating strategic expansion, with companies prioritizing Asia-Pacific scaling, North American compliance integration, and European regulatory-driven innovation.
North America holds approximately 36% of the Industrial Portable Active Microbial Air Samplers Market, driven by concentrated pharmaceutical manufacturing capacity, biologics production expansion, and strict environmental compliance requirements. The United States dominates regional demand with over 2,800 regulated sterile production facilities actively modernizing contamination-control infrastructure. Revised FDA inspection priorities and increasing biologics manufacturing investments accelerated portable monitoring deployment by nearly 24% between 2024 and 2026. Companies are rapidly integrating AI-enabled microbial analytics and cloud-connected validation systems to optimize cleanroom monitoring efficiency and reduce audit preparation time by approximately 28%. Major manufacturers are also expanding regional calibration and servicing networks to improve response speed and lifecycle support. Enterprise buyers increasingly prioritize automated traceability, compliance integration, and operational uptime, making North America the primary region for premium technology deployment and long-term strategic investment.
Europe represents nearly 27% of the Industrial Portable Active Microbial Air Samplers Market, supported by stringent sterile manufacturing regulations and strong environmental monitoring enforcement across Germany, France, Switzerland, and the United Kingdom. Revised EU GMP Annex 1 implementation accelerated advanced portable monitoring adoption by approximately 22%, particularly within pharmaceutical and biologics manufacturing facilities. European enterprises are prioritizing low-intervention digital monitoring systems that reduce manual documentation workloads by nearly 26% while strengthening audit consistency and ESG-aligned operational efficiency. Companies are investing heavily in automated data traceability, wireless contamination monitoring, and energy-efficient cleanroom validation systems. Compliance-first purchasing behavior dominates regional demand, with buyers prioritizing precision, validation reliability, and lifecycle service support over low-cost alternatives. Europe is forcing suppliers to accelerate product innovation and regulatory adaptability, making the region strategically critical for next-generation contamination-control technologies.
Asia-Pacific accounts for approximately 24% of global Industrial Portable Active Microbial Air Samplers Market demand and represents the fastest-expanding regional manufacturing ecosystem. China, India, South Korea, Japan, and Singapore are rapidly increasing pharmaceutical, semiconductor, and biologics production capacity, creating strong demand for portable contamination-control infrastructure. Regional localized manufacturing activity increased nearly 27% between 2024 and 2026, reducing equipment lead times and strengthening supply responsiveness. Enterprises are aggressively adopting wireless portable monitoring systems and digital compliance tools to support high-volume cleanroom operations and modular production expansion. Semiconductor fabrication growth alone pushed airborne contamination monitoring investments up by approximately 21% across advanced manufacturing facilities. Companies operating in Asia-Pacific prioritize scalability, deployment speed, and operational flexibility, making the region essential for production expansion, cost optimization, and long-term global supply chain positioning.
South America contributes nearly 7% of the Industrial Portable Active Microbial Air Samplers Market, with Brazil and Argentina leading demand through expanding pharmaceutical manufacturing and healthcare infrastructure modernization initiatives. Contamination-control investments increased by approximately 18% during 2025 as regional biologics production and sterile processing activities accelerated. However, high import dependency and fluctuating currency conditions continue constraining rapid technology scaling, increasing equipment acquisition costs by nearly 14% in some industrial sectors. Enterprises are responding through phased deployment strategies and localized distributor partnerships focused on affordable portable monitoring solutions. Demand is strongest among mid-sized pharmaceutical facilities seeking flexible compliance systems with lower operational complexity. Regional buyers remain highly price-sensitive while prioritizing durability and service accessibility, positioning South America as a strategic opportunity market balanced against operational and economic execution risk.
The Middle East & Africa region contributes approximately 6% of global Industrial Portable Active Microbial Air Samplers Market demand, led by Saudi Arabia, the UAE, and South Africa through expanding pharmaceutical production, healthcare modernization, and industrial diversification initiatives. Regional cleanroom infrastructure investments increased nearly 20% between 2024 and 2026 as governments accelerated healthcare localization and sterile manufacturing capability development. Enterprises are increasingly deploying portable wireless monitoring systems to support hospital sterilization upgrades and pharmaceutical validation programs. Strategic partnerships with international contamination-control providers improved deployment speed by approximately 17% across newly commissioned facilities. Buyers prioritize operational reliability, scalable deployment, and long-term technical support due to varying infrastructure maturity levels. The region is emerging as a strategic investment destination for suppliers targeting healthcare expansion, industrial modernization, and localized contamination-control ecosystem development.
United States – 34% Market share: Dominates due to extensive sterile pharmaceutical manufacturing capacity, advanced biologics production, and strict FDA environmental monitoring requirements.
China – 16% Market share: Leads demand through rapid pharmaceutical infrastructure expansion, localized cleanroom manufacturing growth, and increasing semiconductor contamination-control investments.
The Industrial Portable Active Microbial Air Samplers Market is dominated by global contamination-control technology leaders competing aggressively against specialized cleanroom monitoring providers and cost-focused regional manufacturers. Key competition centers around Sartorius, Merck, bioMérieux, Particle Measuring Systems, VWR, and MBV AG, with global technology innovators competing directly against regional calibration-focused suppliers and integrated cleanroom solution providers. The top five players collectively control nearly 54% of global market positioning through strong regulatory validation capability, advanced contamination analytics, and broad pharmaceutical customer networks.
Competition is increasingly driven by technological precision, automation integration, compliance traceability, and lifecycle servicing rather than hardware pricing alone. AI-enabled microbial detection systems improve contamination analysis efficiency by nearly 35%, while wireless integrated monitoring platforms reduce compliance reporting workloads by approximately 25%, intensifying pressure on traditional standalone equipment suppliers. Companies are responding through regional expansion, software partnerships, cloud-based monitoring ecosystems, and vertically integrated calibration services. The competitive landscape is rapidly shifting toward intelligent contamination-management platforms as buyers prioritize interoperability and audit-ready digital workflows. High validation barriers, regulatory certification requirements, and precision component dependency continue restricting new entrants. Winning in this market now requires advanced compliance integration, rapid deployment capability, strong servicing infrastructure, and continuous monitoring innovation.
Merck KGaA
bioMérieux SA
MBV AG
Particle Measuring Systems
VWR International
Climet Instruments Company
Lighthouse Worldwide Solutions
Bertin Technologies
EMTEK
Orum International
Cherwell Laboratories
AES Laboratoire
Air Techniques International
Advanced digital contamination-monitoring technologies are redefining the Industrial Portable Active Microbial Air Samplers Market as industrial operators transition toward intelligent environmental monitoring ecosystems. AI-assisted colony recognition systems, wireless IoT-enabled samplers, and automated compliance-reporting platforms are now central to operational modernization across pharmaceutical, biotechnology, and semiconductor cleanrooms. AI-based microbial analytics improve contamination detection efficiency by nearly 34% while reducing manual interpretation workloads by approximately 28%, enabling faster validation and investigation cycles. More than 52% of newly deployed portable microbial monitoring systems now include cloud-connected traceability and automated audit documentation functionality.
Rapid microbial detection technologies are also transforming environmental monitoring speed and operational responsiveness. ATP bioluminescence-based systems and advanced optical detection platforms reduce sampling turnaround time by nearly 38% compared to conventional culture-based monitoring workflows. Portable wireless systems simultaneously improve deployment flexibility and lower cleanroom intervention frequency by approximately 22%, reducing contamination exposure risks during monitoring operations. Companies with advanced biologics production and aseptic manufacturing environments are gaining the strongest operational advantage from these integrated systems.
The competitive gap between legacy manual monitoring and next-generation automated systems is widening significantly. New AI-enabled portable samplers improve monitoring productivity by over 35% while reducing compliance-related labor costs by nearly 24% compared to traditional standalone samplers. Technology leaders are aggressively integrating predictive analytics, automated calibration, and centralized contamination dashboards to strengthen lifecycle service models and customer retention.
Between 2026 and 2028, the market will shift further toward integrated environmental intelligence platforms combining microbial monitoring, particulate tracking, and digital compliance ecosystems. Companies investing early in intelligent portable monitoring infrastructure will secure stronger operational resilience, faster audit readiness, and scalable contamination-control capability across increasingly complex industrial production networks.
May 2025 – Sartorius AG launched the Octet® R8e high-sensitivity biomolecular analysis platform with 384-well compatibility and reduced sample volume requirements down to 40 µL, significantly improving detection throughput and operational efficiency for advanced sterile manufacturing and environmental monitoring workflows. [High-Sensitivity Expansion] Source: www.sartorius.com
April 2025 – bioMérieux introduced the WATCHFIRE™ molecular environmental surveillance solution capable of detecting 22 pathogens through integrated BIOFIRE® FILMARRAY® TORCH instrumentation and FIREWORKS™ software, strengthening real-time contamination and wastewater monitoring capabilities for industrial and public-health environments. [Environmental Surveillance Launch]
September 2025 – bioMérieux reported a 10% increase in Pharma Quality Control segment performance within its Industrial Applications business, driven by expanding contamination-control and environmental monitoring demand across regulated pharmaceutical manufacturing operations globally. [Pharma QC Acceleration]
May 2025 – GXO Logistics and bioMérieux expanded their long-term strategic partnership to optimize instrumentation distribution and supply-chain efficiency, strengthening operational responsiveness and logistics support for advanced diagnostic and contamination-monitoring equipment deployment across Europe. [Supply Chain Optimization]
The Industrial Portable Active Microbial Air Samplers Market Report delivers comprehensive coverage across product types, industrial applications, end-user industries, regional demand centers, and emerging contamination-monitoring technologies shaping advanced cleanroom operations. The report evaluates portable impaction-based systems, slit-to-agar technologies, rapid microbial detection platforms, and integrated wireless monitoring solutions deployed across pharmaceutical manufacturing, biotechnology production, semiconductor fabrication, food processing, healthcare sterilization, and research laboratories. Geographic analysis spans North America, Europe, Asia-Pacific, South America, and the Middle East & Africa with detailed assessment of regional adoption intensity, operational modernization trends, and compliance-driven deployment patterns.
The study incorporates deep analytical benchmarking across more than 25 market-level indicators including deployment concentration, technology adoption rates, contamination-monitoring workflow efficiency, and competitive positioning. Approximately 57% of industrial buyers now prioritize cloud-enabled compliance systems, while AI-assisted monitoring adoption surpassed 30% across advanced sterile manufacturing environments. The report also evaluates evolving demand for rapid microbial detection systems, wireless portable sampling infrastructure, and integrated environmental intelligence platforms expected to reshape contamination-control operations between 2026 and 2033.
Strategically, the report supports investment planning, regional expansion, product development prioritization, supply chain positioning, and competitive benchmarking. It provides decision-makers with execution-level insight into high-growth industrial contamination-control opportunities, emerging operational risks, and technology shifts redefining long-term market leadership.
| Report Attribute / Metric | Details |
|---|---|
| Market Revenue (2025) | USD 59.2 Million |
| Market Revenue (2033) | USD 96.5 Million |
| CAGR (2026–2033) | 6.3% |
| Base Year | 2025 |
| Forecast Period | 2026–2033 |
| Historic Period | 2021–2025 |
| Segments Covered |
By Type
By Application
By End-User Insights
|
| Key Report Deliverables | Revenue Forecast; Market Trends; Growth Drivers & Restraints; Technology Insights; Segmentation Analysis; Regional Insights; Competitive Landscape; Regulatory & ESG Overview; Recent Developments |
| Regions Covered | North America; Europe; Asia-Pacific; South America; Middle East & Africa |
| Key Players Analyzed | Sartorius AG; Merck KGaA; bioMérieux SA; MBV AG; Particle Measuring Systems; VWR International; Climet Instruments Company; Lighthouse Worldwide Solutions; Bertin Technologies; EMTEK; Orum International; Cherwell Laboratories; AES Laboratoire; Air Techniques International |
| Customization & Pricing | Available on Request (10% Customization Free) |
