Reports
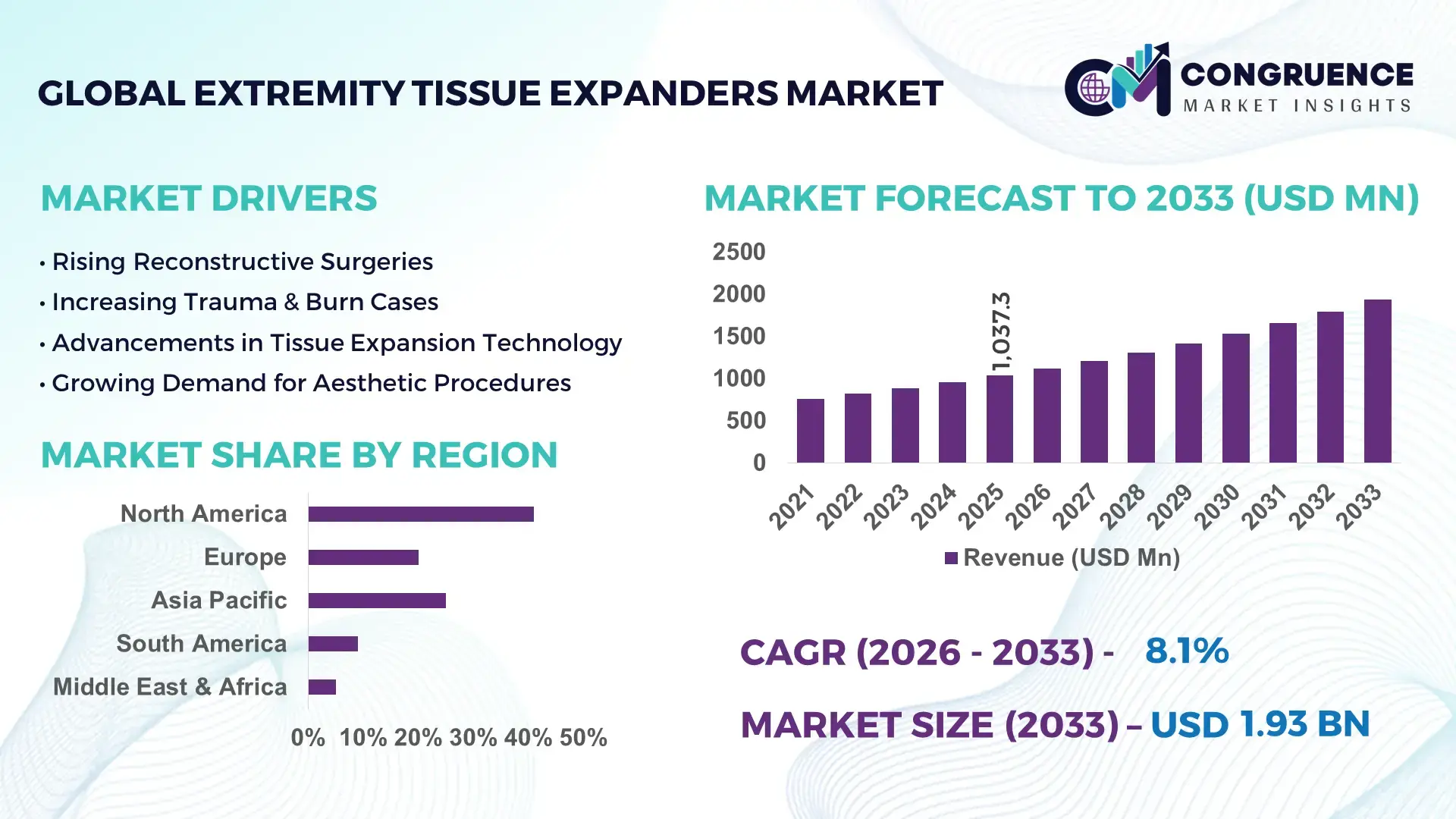
The Global Extremity Tissue Expanders Market was valued at USD 1037.33 Million in 2025 and is anticipated to reach a value of USD 1934.29 Million by 2033 expanding at a CAGR of 8.1% between 2026 and 2033. This growth is primarily driven by the rising demand for advanced reconstructive procedures and increasing incidence of trauma and congenital deformities requiring soft tissue reconstruction.
The United States continues to demonstrate strong operational capacity in the extremity tissue expanders market, supported by over 6,000 accredited surgical centers performing reconstructive and plastic surgeries annually. Investment in advanced biomaterials and implantable medical devices exceeded USD 5 billion in recent years, with extremity reconstruction accounting for a notable share in trauma-related interventions. Adoption rates of advanced tissue expansion systems in orthopedic and reconstructive surgeries exceed 65% across tertiary care hospitals. Additionally, technological integration such as remote-controlled expanders and antibiotic-coated devices has improved surgical outcomes by up to 30% in post-operative recovery metrics. High utilization in burn treatment centers and limb reconstruction programs further strengthens the country’s industrial and clinical ecosystem.
Market Size & Growth: Valued at USD 1037.33 Million in 2025, projected to reach USD 1934.29 Million by 2033 at 8.1% CAGR, driven by increasing reconstructive surgeries and trauma cases.
Top Growth Drivers: Surgical procedure volume growth by 18%, biomaterial efficiency improvement by 25%, and patient-specific implant adoption rising by 32%.
Short-Term Forecast: By 2028, surgical complication rates are expected to reduce by 20% due to advanced expander technologies.
Emerging Technologies: Smart tissue expanders with remote monitoring, biodegradable expanders, and 3D-printed patient-specific implants.
Regional Leaders: North America projected at USD 720 Million by 2033 with high clinical adoption; Europe at USD 520 Million driven by regulatory-backed innovation; Asia-Pacific at USD 480 Million with expanding surgical infrastructure.
Consumer/End-User Trends: Hospitals account for over 60% usage, with increasing adoption among specialized reconstructive clinics and ambulatory surgical centers.
Pilot or Case Example: In 2024, a clinical pilot demonstrated a 28% reduction in recovery time using sensor-enabled expanders in limb reconstruction procedures.
Competitive Landscape: Market leader holds approximately 28% share, followed by major players focusing on biomaterial innovation and device customization.
Regulatory & ESG Impact: Increasing compliance with biocompatibility standards and adoption of recyclable medical-grade polymers.
Investment & Funding Patterns: Over USD 900 Million invested in reconstructive device innovation and surgical robotics integration.
Innovation & Future Outlook: Integration of AI-assisted surgical planning and minimally invasive expansion systems shaping next-generation solutions.
The extremity tissue expanders market demonstrates strong alignment with orthopedic surgery, plastic reconstruction, and trauma care sectors, with hospitals contributing over 60% of procedural demand while specialty clinics account for approximately 25%. Innovations such as silicone-based expandable implants with antimicrobial coatings and pressure-controlled inflation systems are improving patient safety and surgical precision. Regulatory frameworks emphasizing biocompatibility and long-term implant safety are accelerating product approvals. Regionally, Asia-Pacific is witnessing rising consumption due to expanding healthcare infrastructure and increasing surgical awareness, while Europe focuses on sustainable material adoption. Future trends include personalized expanders, AI-integrated surgical workflows, and enhanced outpatient procedures, all contributing to improved efficiency and reduced hospitalization durations.
The extremity tissue expanders market holds strategic relevance within the broader reconstructive surgery ecosystem, driven by the increasing need for precision-based soft tissue regeneration in trauma, oncology, and congenital deformity treatments. Healthcare systems are prioritizing advanced reconstructive solutions that reduce hospitalization time and improve patient outcomes. Smart tissue expanders equipped with sensor-based monitoring deliver approximately 35% improvement in inflation accuracy compared to conventional manual expansion systems, significantly enhancing procedural safety and predictability.
North America dominates in procedural volume due to well-established surgical infrastructure, while Asia-Pacific leads in adoption growth with over 40% of healthcare facilities integrating advanced reconstructive technologies. By 2028, AI-assisted surgical planning is expected to improve pre-operative accuracy by nearly 30%, enabling surgeons to optimize expansion timelines and minimize complications. Additionally, biodegradable expander technologies are projected to reduce secondary removal procedures by 25%, enhancing patient convenience and lowering healthcare costs.
From an ESG and compliance perspective, manufacturers are committing to reducing medical-grade polymer waste by 20% by 2030, alongside adopting recyclable and biocompatible materials. Regulatory bodies are enforcing stringent safety benchmarks, accelerating the shift toward next-generation implantable devices with reduced infection risks. In 2024, a leading medical device initiative in Germany achieved a 22% reduction in post-surgical complications through the implementation of digitally controlled expansion systems. Strategically, the extremity tissue expanders market is evolving as a critical enabler of precision medicine and value-based healthcare delivery. Its integration with digital health technologies, minimally invasive procedures, and sustainable manufacturing practices positions it as a foundational pillar for resilient, compliant, and future-ready surgical ecosystems.
The increasing incidence of trauma-related injuries and reconstructive surgical procedures is a primary driver of the extremity tissue expanders market. Globally, over 50 million individuals sustain injuries requiring medical attention annually, with a substantial proportion needing soft tissue reconstruction. Burn injuries alone account for approximately 11 million cases each year, many of which require staged tissue expansion procedures. Hospitals are reporting a 20–25% increase in reconstructive surgeries involving limbs due to improved access to healthcare and surgical advancements. The demand for effective wound closure and tissue regeneration solutions is further amplified by the rising number of orthopedic procedures and limb salvage surgeries. Advanced tissue expanders enable surgeons to generate additional skin with similar texture and vascularity, improving surgical outcomes by nearly 30%. This growing clinical reliance on tissue expansion technologies continues to strengthen market demand across developed and emerging healthcare systems.
Despite technological advancements, high procedural costs associated with extremity tissue expanders remain a significant barrier to widespread adoption. The overall cost of tissue expansion procedures, including device costs, surgical fees, and post-operative care, can be 25–40% higher than alternative reconstructive methods. In low- and middle-income regions, limited healthcare funding and inadequate reimbursement policies restrict patient access to advanced reconstructive treatments. Additionally, the requirement for multiple clinical visits for gradual expansion increases the financial and logistical burden on patients. Skilled surgical expertise is also concentrated in urban healthcare centers, leaving rural populations underserved. Data indicates that less than 35% of eligible patients in developing regions receive advanced reconstructive procedures due to cost and infrastructure limitations. These economic and accessibility challenges continue to hinder the full-scale adoption of extremity tissue expanders across global markets.
The integration of personalized and smart implant technologies presents significant growth opportunities in the extremity tissue expanders market. The adoption of 3D printing in medical device manufacturing has increased by over 40%, enabling the production of patient-specific expanders tailored to anatomical requirements. These customized solutions improve surgical precision and reduce complications by approximately 20%. Additionally, smart expanders equipped with pressure sensors and remote monitoring capabilities are gaining traction, allowing clinicians to track expansion progress in real time and minimize manual interventions. The rise of outpatient surgical procedures and ambulatory care centers further expands the potential for minimally invasive tissue expansion solutions. Emerging markets are investing heavily in healthcare digitization, creating opportunities for AI-integrated surgical planning and predictive analytics. These technological advancements are expected to enhance patient outcomes, reduce hospital stays, and create new revenue streams for manufacturers and healthcare providers.
The extremity tissue expanders market faces ongoing challenges related to regulatory complexity and post-surgical complications. Medical devices used for tissue expansion must comply with stringent safety and biocompatibility standards, often requiring extensive clinical validation and approval timelines that can exceed 3–5 years. Variations in regulatory frameworks across regions further complicate global product commercialization. Post-surgical risks such as infection, device leakage, and tissue necrosis remain concerns, with complication rates ranging between 10% and 15% in certain procedures. These risks necessitate additional monitoring and sometimes revision surgeries, increasing healthcare costs and affecting patient confidence. Furthermore, maintaining consistent product quality while incorporating advanced features such as sensors and biodegradable materials presents manufacturing challenges. Addressing these issues requires continuous investment in research, quality assurance, and regulatory compliance, which can strain smaller market participants and slow overall innovation.
• Accelerated Adoption of Smart and Sensor-Enabled Expanders Improving Clinical Precision by 35%
The integration of smart technologies into extremity tissue expanders is significantly transforming surgical outcomes, with sensor-enabled devices improving inflation accuracy by nearly 35% compared to traditional systems. These advanced expanders allow real-time pressure monitoring and automated adjustments, reducing manual intervention frequency by approximately 40%. Hospitals adopting digital monitoring systems have reported a 25% reduction in post-operative complications and a 20% improvement in patient recovery timelines. Additionally, over 50% of advanced reconstructive centers in North America have incorporated remote-controlled expanders into their surgical protocols. This trend is particularly impactful in complex limb reconstruction cases where precision and controlled expansion are critical to avoid tissue damage and ensure optimal vascularization.
• Rising Demand for Biodegradable and Antimicrobial Materials Enhancing Safety by 30%
Material innovation is emerging as a key trend, with biodegradable and antimicrobial-coated tissue expanders gaining traction across healthcare systems. These advanced materials have demonstrated up to 30% reduction in infection rates and eliminate the need for secondary removal procedures in nearly 20% of cases. Approximately 45% of newly developed expanders now incorporate antimicrobial coatings, improving patient safety and long-term outcomes. In Europe, over 35% of hospitals have transitioned toward eco-friendly and biocompatible implant solutions aligned with stringent regulatory standards. This shift is also driven by sustainability goals, with manufacturers targeting a 25% reduction in non-recyclable medical waste by adopting next-generation polymer technologies.
• Expansion of Outpatient and Minimally Invasive Procedures Increasing Adoption by 28%
The shift toward outpatient care and minimally invasive surgical procedures is reshaping the extremity tissue expanders market. Data indicates that nearly 28% of tissue expansion procedures are now performed in ambulatory surgical centers, reducing hospital stays by up to 40%. Minimally invasive techniques utilizing smaller incisions and advanced expanders have lowered procedural risks by approximately 22%. In Asia-Pacific, outpatient surgical adoption has grown by 33% over recent years, supported by expanding healthcare infrastructure and cost-effective treatment models. This trend is enabling broader patient access while improving operational efficiency for healthcare providers, particularly in urban and semi-urban regions.
• Increasing Use of 3D Printing and Customization Driving Patient-Specific Solutions by 40%
Customization through 3D printing technology is rapidly gaining momentum, with over 40% of advanced healthcare facilities now utilizing patient-specific tissue expanders tailored to anatomical requirements. These customized solutions have shown a 20–25% improvement in surgical precision and a 15% reduction in complication rates. The adoption of digital modeling and pre-surgical simulation tools has increased by 38%, enabling surgeons to design optimized expansion pathways. In the United States and Japan, more than 30% of complex reconstructive procedures now incorporate 3D-printed expanders. This trend is driving innovation in personalized medicine, improving patient outcomes, and enhancing the efficiency of reconstructive surgical workflows.
The extremity tissue expanders market is segmented based on type, application, and end-user, each contributing uniquely to overall industry performance. Silicone-based expanders dominate the product landscape due to their durability and biocompatibility, while emerging biodegradable variants are gaining traction in advanced healthcare systems. In terms of application, reconstructive surgery accounts for the majority of usage, particularly in trauma and burn cases, followed by cosmetic procedures and congenital defect correction. Hospitals represent the largest end-user segment, driven by access to specialized surgical infrastructure and skilled professionals, while ambulatory surgical centers are expanding rapidly due to cost efficiency and shorter recovery timelines. Regional segmentation highlights North America and Europe as mature markets with high adoption rates, while Asia-Pacific demonstrates strong growth potential due to increasing healthcare investments and rising surgical volumes.
Silicone-based extremity tissue expanders remain the leading product category, accounting for approximately 58% of total adoption due to their proven durability, flexibility, and compatibility with long-term implantation procedures. Saline-filled expanders hold around 27% share, offering controlled expansion capabilities and ease of adjustment during staged procedures. However, biodegradable tissue expanders represent the fastest-growing segment, expanding at an estimated CAGR of 9.4%, driven by their ability to eliminate secondary removal surgeries and reduce patient discomfort by nearly 20%. Hybrid expanders incorporating antimicrobial coatings and smart sensors contribute to the remaining 15% share, catering to specialized surgical needs. The increasing demand for infection-resistant and patient-specific solutions is accelerating innovation across all product categories.
Reconstructive surgery dominates the application segment, accounting for approximately 64% of total usage, primarily driven by trauma cases, burn injuries, and post-oncological reconstruction procedures. Cosmetic and aesthetic applications contribute around 21%, supported by rising demand for minimally invasive procedures and improved aesthetic outcomes. Congenital defect correction and limb deformity treatments collectively represent about 15% of the market. While reconstructive applications remain dominant, cosmetic procedures are the fastest-growing segment, with an estimated CAGR of 8.7%, fueled by increasing patient awareness and advancements in surgical techniques. The integration of digital planning tools and precision expanders is further enhancing procedural success rates across applications.
In 2025, a global healthcare initiative reported that advanced tissue expansion techniques were implemented in over 120 specialized burn care centers, improving reconstructive success rates by nearly 25% and significantly enhancing patient recovery outcomes in complex cases.
Hospitals lead the end-user segment, accounting for approximately 62% of total market utilization due to their comprehensive surgical infrastructure, availability of specialized professionals, and access to advanced medical technologies. Ambulatory surgical centers represent around 23% of usage, benefiting from cost-efficient procedures and reduced patient recovery times. Specialty clinics and reconstructive centers contribute the remaining 15%, focusing on niche and high-precision surgical interventions. Among these, ambulatory surgical centers are the fastest-growing segment, with an estimated CAGR of 9.1%, driven by increasing preference for outpatient procedures and operational efficiency improvements of nearly 30%. The adoption of advanced tissue expanders in these facilities has improved patient throughput and reduced hospitalization dependency.
Region North America accounted for the largest market share at 41% in 2025 however, Asia-Pacific is expected to register the fastest growth, expanding at a CAGR of 9.6% between 2026 and 2033.
North America recorded over 520,000 reconstructive procedures annually involving tissue expansion techniques, supported by more than 6,500 specialized surgical facilities. Europe followed with a 29% share, driven by strong regulatory frameworks and adoption of biocompatible materials across over 3,800 hospitals. Asia-Pacific held approximately 21% share in 2025, with surgical volumes exceeding 1.2 million procedures annually across China, India, and Japan, reflecting rapid infrastructure expansion. South America accounted for nearly 5% share, with Brazil contributing over 55% of regional procedures, while Middle East & Africa held around 4%, driven by increasing investments in healthcare facilities and rising demand for advanced reconstructive treatments. Across regions, adoption of smart expanders has grown by over 30%, and biodegradable implants are being integrated in more than 25% of newly approved medical devices globally.
How are advanced surgical ecosystems and digital innovation accelerating adoption of reconstructive technologies?
North America dominates the extremity tissue expanders market with approximately 41% share, supported by a highly developed healthcare infrastructure and strong demand across trauma care, plastic surgery, and oncology reconstruction. The region performs over 500,000 limb reconstruction and soft tissue procedures annually, with hospitals accounting for nearly 65% of device utilization. Regulatory support from agencies enforcing strict biocompatibility and safety standards has accelerated the approval of next-generation expanders. Technological advancements such as sensor-enabled expanders and AI-assisted surgical planning have improved procedural efficiency by nearly 30%. A key regional player has expanded its portfolio of antimicrobial-coated expanders, contributing to a 22% reduction in post-operative infection rates. Consumer behavior reflects a strong preference for advanced, minimally invasive procedures, with over 60% of patients opting for outpatient surgical solutions when available.
Why is regulatory-driven innovation shaping the demand for sustainable and advanced reconstructive solutions?
Europe holds around 29% of the extremity tissue expanders market, with key countries such as Germany, the United Kingdom, and France contributing significantly to regional demand. Over 3,500 healthcare facilities across Europe actively perform reconstructive procedures using advanced tissue expansion technologies. Stringent regulatory bodies emphasizing patient safety and environmental sustainability have driven the adoption of biodegradable and recyclable implant materials, now used in nearly 35% of procedures. Emerging technologies such as 3D printing and digital surgical planning tools have increased precision rates by approximately 25%. A regional manufacturer has introduced eco-friendly expanders that reduce medical waste by 20%, aligning with EU sustainability goals. Consumer behavior in this region shows a strong inclination toward safe, compliant, and environmentally responsible medical devices, influencing purchasing decisions across healthcare providers.
What factors are driving rapid expansion of reconstructive healthcare infrastructure and surgical adoption?
Asia-Pacific represents the fastest-growing region in the extremity tissue expanders market, accounting for approximately 21% of global volume. Countries such as China, India, and Japan collectively perform over 1 million reconstructive procedures annually, supported by expanding healthcare infrastructure and increasing medical tourism. The region has witnessed a 35% increase in the number of specialized surgical centers over recent years, enhancing accessibility to advanced treatments. Manufacturing capabilities are also strengthening, with local production of medical-grade implants increasing by 28%, reducing dependency on imports. Technological innovation hubs in Japan and South Korea are leading the development of smart expanders and AI-integrated surgical systems. A regional manufacturer has introduced cost-effective expansion devices, improving accessibility for nearly 40% of mid-income patients. Consumer behavior is characterized by rising awareness and preference for affordable, high-quality surgical solutions, particularly in urban areas.
How are healthcare investments and policy reforms influencing reconstructive surgery adoption?
South America accounts for approximately 5% of the extremity tissue expanders market, with Brazil and Argentina emerging as key contributors. Brazil alone performs over 120,000 reconstructive procedures annually, supported by government initiatives aimed at expanding healthcare access. Infrastructure development in urban healthcare centers has increased surgical capacity by nearly 18% over the past few years. Trade policies supporting medical device imports have improved availability of advanced expanders, while local manufacturers are gradually increasing production capabilities. A regional company has focused on developing cost-efficient expanders, reducing procedural costs by approximately 15%. Consumer behavior in this region reflects a growing demand for reconstructive and cosmetic procedures, particularly among middle-income populations, with increased awareness contributing to a 20% rise in elective surgeries.
What role do healthcare modernization and strategic partnerships play in advancing surgical capabilities?
The Middle East & Africa region holds approximately 4% share in the extremity tissue expanders market, with countries such as the UAE and South Africa leading adoption. The region has seen a 25% increase in healthcare infrastructure investments, including the establishment of advanced surgical centers and specialized clinics. Technological modernization initiatives have introduced minimally invasive procedures and digital surgical planning tools, improving surgical outcomes by nearly 20%. Trade partnerships with global medical device manufacturers have enhanced access to high-quality implants and advanced technologies. A regional healthcare provider has implemented advanced reconstructive programs, increasing procedure success rates by 18%. Consumer behavior is evolving, with rising demand for high-quality medical treatments and increasing medical tourism contributing to higher adoption rates of extremity tissue expanders.
United States – 38% share in the Extremity Tissue Expanders market, driven by advanced healthcare infrastructure and high volume of reconstructive surgeries.
Germany – 12% share in the Extremity Tissue Expanders market, supported by strong regulatory frameworks and widespread adoption of innovative medical technologies.
The extremity tissue expanders market exhibits a moderately consolidated competitive structure, with the top five companies accounting for approximately 62% of the global market share. Over 25 active global and regional players are competing through continuous innovation, strategic partnerships, and product differentiation. Market leaders are heavily investing in research and development, allocating nearly 12–15% of their annual budgets toward advanced biomaterials, smart expanders, and minimally invasive solutions. Product innovation remains a key competitive factor, with over 40% of newly launched devices incorporating antimicrobial coatings or digital monitoring capabilities. Strategic collaborations between medical device manufacturers and healthcare institutions have increased by 30%, enabling faster clinical validation and product adoption. Mergers and acquisitions activity has also intensified, with at least 8 major deals recorded in recent years to strengthen product portfolios and geographic presence. Additionally, companies are focusing on expanding their footprint in emerging markets, where demand for cost-effective and technologically advanced solutions is rising. Competitive differentiation is increasingly driven by customization capabilities, regulatory compliance, and integration of digital health technologies.
Mentor Worldwide LLC
Allergan Aesthetics
Sientra Inc.
GC Aesthetics
PMT Corporation
Polytech Health & Aesthetics GmbH
Wanhe Plastic Materials Co., Ltd.
Groupe Sebbin SAS
Koken Co., Ltd.
Nagor Ltd.
Technological advancements in the extremity tissue expanders market are centered on improving surgical precision, patient safety, and procedural efficiency. One of the most impactful innovations is the integration of sensor-enabled and digitally controlled expanders, which provide real-time monitoring of pressure levels and tissue response. These systems have demonstrated up to 35% improvement in expansion accuracy and reduced manual intervention by nearly 40%, enabling clinicians to optimize treatment protocols. Wireless and remote-controlled expanders are also gaining traction, with adoption increasing by over 30% across advanced surgical centers, allowing gradual and controlled tissue expansion without repeated invasive procedures.
Material science innovations are playing a crucial role, particularly the development of next-generation silicone elastomers and biodegradable polymers. Advanced silicone-based expanders now exhibit up to 25% higher durability and reduced rupture rates, while biodegradable variants eliminate the need for secondary removal surgeries in approximately 20% of cases. Antimicrobial coatings are now incorporated in over 45% of newly developed expanders, reducing infection risks by nearly 30% and improving post-operative outcomes. These coatings are particularly beneficial in trauma and burn reconstruction, where infection control is critical.
Additive manufacturing and 3D printing technologies are transforming customization capabilities within the market. Over 40% of high-complexity reconstructive procedures now utilize patient-specific expanders designed using digital imaging and modeling tools. This approach enhances anatomical fit and improves surgical outcomes by 20–25%. AI-assisted surgical planning platforms are further enhancing pre-operative precision, reducing planning time by nearly 35% and improving procedural success rates.
Minimally invasive surgical techniques are also evolving, supported by smaller, flexible expanders that reduce incision size by up to 30% and decrease recovery time by approximately 25%. Integration with digital health platforms enables data-driven decision-making, with hospitals reporting a 28% improvement in patient monitoring efficiency. Collectively, these technological advancements are redefining clinical standards and enabling more predictable, efficient, and patient-centric reconstructive procedures.
• In April 2025, Sientra Inc. announced the expansion of its reconstructive product portfolio with enhanced tissue expander designs featuring improved shell strength and integrated antimicrobial protection, aimed at reducing complication rates and improving patient outcomes in complex extremity reconstruction procedures. Source: www.sientra.com
• In September 2024, GC Aesthetics introduced a new generation of anatomical tissue expanders incorporating advanced silicone gel technology and improved valve systems, designed to enhance expansion control and reduce leakage risks during staged reconstructive surgeries. Source: www.gcaesthetics.com
• In February 2025, Polytech Health & Aesthetics GmbH expanded its production capabilities in Europe, increasing output of medical-grade silicone implants and expanders by over 20% to meet rising demand for reconstructive procedures and ensure consistent product availability. Source: www.polytechhealth.com
• In November 2024, Mentor Worldwide LLC launched an updated line of tissue expanders with refined surface texturing and enhanced biocompatibility features, targeting improved integration with surrounding tissue and reduced post-operative complications in reconstructive surgeries. Source: www.mentorwwllc.com
The extremity tissue expanders market report provides a comprehensive evaluation of industry performance across multiple dimensions, including product types, clinical applications, end-user segments, and geographic distribution. The report covers key product categories such as silicone-based, saline-filled, biodegradable, and hybrid expanders, which collectively account for over 95% of market utilization. It also assesses technological advancements, including sensor-enabled expanders, antimicrobial coatings, and 3D-printed customized devices, which are increasingly integrated into over 40% of advanced surgical procedures.
From an application perspective, the report analyzes reconstructive surgery, cosmetic procedures, and congenital defect correction, with reconstructive applications contributing to more than 60% of overall usage. End-user segmentation includes hospitals, ambulatory surgical centers, and specialty clinics, with hospitals representing approximately 62% of total demand due to their access to specialized infrastructure and skilled professionals. The report further evaluates emerging outpatient care models, which now account for nearly 28% of procedures globally.
Geographically, the report spans North America, Europe, Asia-Pacific, South America, and the Middle East & Africa, capturing regional variations in adoption, infrastructure, and technological integration. North America leads with over 40% share, while Asia-Pacific demonstrates strong growth potential with increasing surgical volumes exceeding 1 million procedures annually. The scope also includes analysis of regulatory frameworks, sustainability initiatives, and innovation trends shaping the industry. Additionally, niche segments such as smart expanders, biodegradable implants, and AI-assisted surgical planning are explored, highlighting their growing influence on market dynamics and future opportunities for stakeholders.
| Report Attribute/Metric | Report Details |
|---|---|
|
Market Revenue in 2025 |
USD V2025 Million |
|
Market Revenue in 2033 |
USD V2033 Million |
|
CAGR (2026 - 2033) |
8.1% |
|
Base Year |
2025 |
|
Forecast Period |
2026 - 2033 |
|
Historic Period |
2021 - 2025 |
|
Segments Covered |
By Types
By Application
By End-User
|
|
Key Report Deliverable |
Revenue Forecast, Growth Trends, Market Dynamics, Segmental Overview, Regional and Country-wise Analysis, Competition Landscape |
|
Region Covered |
North America, Europe, Asia-Pacific, South America, Middle East, Africa |
|
Key Players Analyzed |
Mentor Worldwide LLC, Allergan Aesthetics, Sientra Inc., GC Aesthetics, PMT Corporation, Polytech Health & Aesthetics GmbH, Wanhe Plastic Materials Co., Ltd., Groupe Sebbin SAS, Koken Co., Ltd., Nagor Ltd. |
|
Customization & Pricing |
Available on Request (10% Customization is Free) |
