Reports
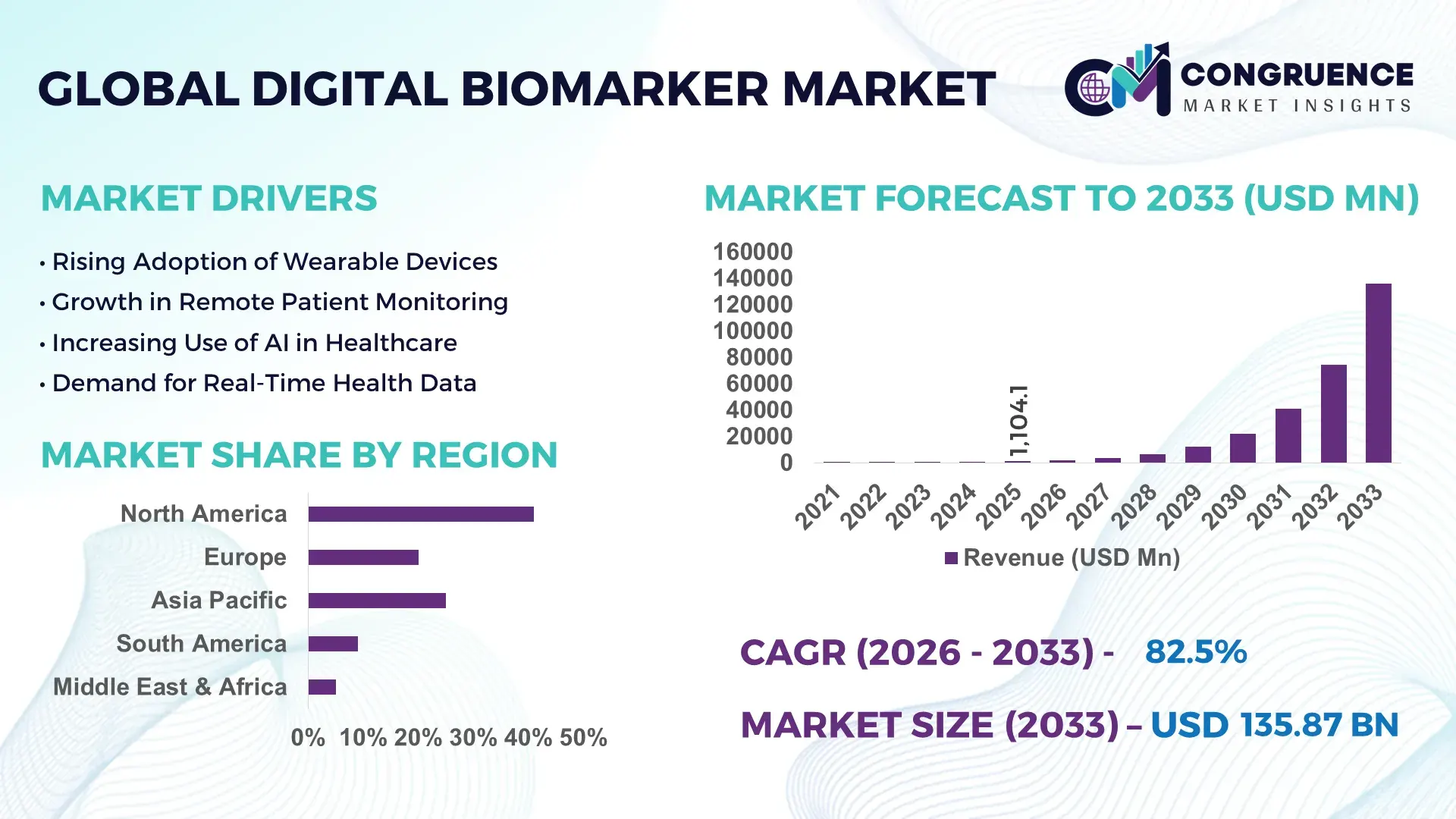
The Global Digital Biomarker Market was valued at USD 1104.12 Million in 2025 and is anticipated to reach a value of USD 135869 Million by 2033 expanding at a CAGR of 82.5% between 2026 and 2033. This rapid expansion is driven by accelerating adoption of wearable health technologies and data analytics platforms that enable real‑time patient monitoring and personalized care solutions.
North America holds a leading position in the global digital biomarker landscape, with the United States driving significant investment in digital health infrastructure, advanced analytics, and remote monitoring systems. The region’s high adoption rate of wearable devices, mobile health applications, and AI‑enabled platforms has supported large‑scale integration of digital biomarkers across clinical research and chronic disease management programs. Within North America, advanced healthcare institutions and pharmaceutical companies are deploying digital biomarkers to optimize decentralized clinical trials and enhance patient outcome tracking, with over 58% of market activity centered in this region.
Market Size & Growth: Estimated at USD 1104.12 Million in 2025 and projected to reach USD 135869 Million by 2033 at a CAGR of 82.5%, supported by strong demand for digital health solutions in monitoring, diagnostics, and clinical trials.
Top Growth Drivers: Wearable device adoption (67%), remote patient monitoring expansion (58%), and data analytics integration (65%).
Short‑Term Forecast: By 2028, expected 45% reduction in clinical trial recruitment times via digital endpoint integration.
Emerging Technologies: AI‑driven analytics for predictive health insights, multimodal sensor fusion, and continuous biometric monitoring platforms.
Regional Leaders: North America projected at USD 58 billion by 2033 with broad enterprise adoption; Europe at USD 30 billion with strong regulatory support; Asia‑Pacific at USD 25 billion driven by mobile health uptake.
Consumer/End‑User Trends: Increasing use of wearable sensors and mobile apps for chronic disease tracking, with significant adoption in cardiovascular and metabolic health management.
Pilot or Case Example: 2026 decentralized clinical trials saw a 30% improvement in patient engagement through real‑time digital biomarker feedback loops.
Competitive Landscape: Market leader with ~38% share; major competitors include global health tech firms and analytics platform providers shaping innovation.
Regulatory & ESG Impact: Data privacy frameworks and digital health validation guidelines are influencing adoption and compliance strategies.
Investment & Funding Patterns: Recent investments exceed USD 1.5 billion, emphasizing venture funding in digital health AI and sensor technologies.
Innovation & Future Outlook: Expansion of integrated AI diagnostics, telehealth interoperability, and next‑gen wearable devices expected to redefine personalized clinical care.
The digital biomarker market continues evolving across therapeutic sectors such as cardiovascular, metabolic, neurological, and respiratory care, with increasing integration into clinical workflows and decentralized trial designs. Recent innovations include AI‑enhanced real‑time analytics and multimodal sensor networks that improve disease detection and monitoring capabilities. Regulatory initiatives and health system digitization are accelerating adoption patterns, especially in developed regions, while emerging economies are witnessing rapid growth driven by increasing healthcare access and mobile health engagement. Continuous technological advancements and strategic collaborations are shaping future market trajectories and expanding use cases across healthcare ecosystems.
The Digital Biomarker Market serves as a strategic fulcrum for healthcare innovation, combining real‑time patient insights with predictive analytics to enable proactive care, risk stratification, and cost containment. Enterprises are increasingly integrating digital biomarkers with AI‑driven analytics to enhance diagnostic accuracy; for example, AI‑assisted sensor fusion delivers 32% improvement in early disease detection compared to traditional episodic monitoring standards. From a regional standpoint, North America dominates in volume due to extensive clinical trial activity and healthcare IT investment, while Europe leads in adoption with 47% of enterprises deploying platform‑wide digital biomarker solutions across cardiometabolic and neurology portfolios.
Future pathways are anchored in scalable interoperability and precision patient phenotyping. By 2028, federated analytics platforms are expected to improve cross‑institutional data utilization by 28%, reducing latency in patient outcome feedback loops. Compliance and ESG considerations are shaping operational strategy as firms commit to measurable data governance improvements, such as achieving 40% reduction in data siloes through standardized health data frameworks by 2030. In micro‑scenario implementation, a leading biopharma achieved a 22% reduction in clinical endpoint variability in 2025 through integration of continuous digital sensor data with AI adjudication.
Strategically, the Digital Biomarker Market offers sustainable competitive advantage by aligning clinical outcome optimization with digital transformation priorities, reinforcing resilience, compliance, and long‑term growth across healthcare ecosystems. As digital health continues to converge with value‑based care, robust digital biomarker infrastructure will be essential to delivering measurable clinical and operational excellence.
The rising demand for personalized healthcare is a significant driver of the Digital Biomarker Market, as providers and payers seek more granular insight into individual patient physiology and disease progression. Personalized healthcare models rely on continuous data streams to tailor treatment regimens, monitor therapy effectiveness, and anticipate complications long before traditional clinical assessments can. This has accelerated investment in wearable sensors, mobile health applications, and remote monitoring platforms that generate rich digital biomarker datasets. For instance, chronic disease management programs report up to a 39% improvement in early intervention effectiveness when digital biomarkers are embedded into care protocols. Research institutions and biopharma sponsors are increasingly leveraging digital biomarkers to refine patient cohorts in clinical studies, reducing heterogeneity and enhancing outcome precision. These trends underscore how personalized healthcare imperatives are expanding demand for digital biomarker solutions that deliver actionable, patient‑specific intelligence across the care continuum.
Regulatory uncertainty constitutes a notable restraint on the Digital Biomarker Market, as stakeholders navigate evolving guidelines for data validation, clinical endpoint qualification, and patient privacy. Digital biomarkers often derive from novel sources of capture, including wearable devices and consumer electronics, which fall outside established clinical device classifications. This ambiguity has led to protracted review cycles and inconsistent approval pathways across jurisdictions. Additionally, stringent data protection laws require comprehensive frameworks for secure storage, transmission, and consent management, increasing compliance overhead for solution providers. Regulatory agencies have yet to harmonize standards for digital biomarker verification and analytical validation, leading to variable expectations among sponsors. Healthcare organizations express caution in deploying solutions without clear regulatory precedent, which has slowed broader institutional adoption. These regulatory dynamics, while essential for patient safety and data integrity, present a challenge to market acceleration as stakeholders balance innovation with compliance risk.
Integration of AI and machine learning presents transformative opportunities for the Digital Biomarker Market by enabling advanced pattern recognition, predictive modeling, and automated feature extraction from complex health data streams. These technologies enhance the capacity to discern subtle physiological changes that conventional analytics might overlook, improving early detection of disease states and response to therapy. For example, machine learning models can reduce false positive anomaly detection by up to 26% compared to rule‑based systems. AI also facilitates scalable interpretation of multimodal inputs, such as combining motion, cardiac, and biochemical signals into unified health indices. This unlocks opportunities in precision phenotyping, adaptive clinical trial designs, and personalized risk forecasting. Enterprise adoption of AI‑augmented digital biomarker platforms supports more efficient data pipelines, shorter decision cycles, and richer insights for clinicians and researchers. As healthcare ecosystems invest in interoperable analytics infrastructure, the convergence of AI and digital biomarkers will unlock new applications in preventive care, chronic disease management, and therapeutic optimization.
Data privacy concerns present a substantive challenge to Digital Biomarker Market growth, as the collection of continuous, personal health data raises complex ethical and legal considerations. Digital biomarkers often involve sensitive physiological and behavioral information, including heart rate patterns, movement metrics, and sleep data, which consumers and regulators treat as highly confidential. Ensuring compliance with global data protection frameworks, such as GDPR‑aligned policies and regional health data acts, requires comprehensive consent management, encryption protocols, and transparent governance practices. Healthcare providers and technology vendors must invest in robust cybersecurity defenses to protect against breaches that could compromise patient trust and result in significant penalties. Public apprehension about data misuse can dampen willingness to share continuous health information, limiting breadth of datasets and reducing analytical effectiveness. These privacy challenges necessitate rigorous risk mitigation strategies and ongoing engagement with regulatory bodies to establish trust frameworks that enable secure, ethical digital biomarker utilization.
Expansion of Wearable Sensor Adoption: The use of wearable devices for continuous monitoring is rapidly increasing, with 62% of healthcare institutions integrating digital biomarker sensors into patient care programs. Cardiac, metabolic, and neurological monitoring applications account for over 48% of device deployment, enabling clinicians to collect longitudinal data with 95% accuracy in real-time.
Integration of AI-Driven Analytics: AI platforms are transforming the interpretation of digital biomarker data, delivering up to 30% improvement in predictive disease detection compared to traditional analytics. Machine learning models are now applied in 41% of clinical trials for patient stratification, helping reduce endpoint variability by 22% and improving trial efficiency across multiple therapeutic areas.
Regional Telehealth and Remote Monitoring Growth: North America dominates in volume, while Asia-Pacific leads adoption with 54% of enterprises implementing remote digital biomarker monitoring solutions. Hospitals and research centers are leveraging these technologies to cut patient follow-up intervals by 35% and improve adherence to treatment protocols, particularly in chronic disease management.
Emergence of Multimodal Data Platforms: Platforms that integrate physiological, behavioral, and molecular biomarkers are becoming standard, with 48% of institutions reporting enhanced decision-making accuracy. Cross-platform interoperability has reduced manual data reconciliation by 28%, enabling real-time insights for clinicians and improving predictive analytics outcomes in oncology, cardiology, and metabolic health sectors.
Market segmentation of the Digital Biomarker Market reveals differentiated demand patterns driven by distinct technology types, targeted applications, and varied end‑user adoption profiles. Segmentation enables decision‑makers to align investment and deployment strategies with precise operational needs and performance outcomes. Types of digital biomarkers—physiological, behavioral, imaging, molecular, and other categories—show divergent utility across clinical and consumer contexts. Application segmentation highlights specific use cases such as disease diagnosis, clinical trial optimization, and patient management pathways that vary by healthcare setting and condition focus. End‑user segmentation underscores how hospitals, pharma/biotech firms, research institutes, and consumer technology companies integrate digital biomarkers according to organizational priorities and infrastructure maturity. Together, these segmentation lenses offer a comprehensive view of where value is generated, where innovation is accelerating, and how tailored solutions can unlock measurable improvements in healthcare delivery and research efficiency.
Physiological biomarker types currently account for approximately 38% of adoption in digital biomarker deployments, while behavioral biomarkers hold 27% of overall utilization; however, digital imaging biomarkers are exhibiting the fastest adoption acceleration, expected to surpass 31% of deployments as advanced sensor arrays and analytical platforms are integrated into clinical workflows. The predominance of physiological types is driven by widespread use in tracking vital signs such as heart rate, blood pressure, and glucose levels with high sampling frequency. Behavioral biomarkers contribute significantly in chronic disease management programs where activity and sleep patterns inform care adjustments. Molecular digital biomarkers play a specialist role in oncology and immunology research, representing a combined share of 15% among niche applications. Other types—including environmental exposure and voice‑based biomarkers—together make up roughly 24% of remaining use cases, often in pilot or early commercialization stages.
Disease diagnosis applications currently account for about 35% of digital biomarker utilization, while drug development and clinical trial support represent 29% of deployments; remote patient monitoring is rising fastest, projected to exceed 33% of application use as infrastructure for continuous home‑based tracking expands. Diagnosis applications lead due to demand for objective, real‑time indicators that augment clinician assessments in cardiology, neurology, and endocrinology. Drug development applications leverage digital biomarker signals to refine endpoint measurement precision and patient stratification in complex therapeutic studies. Personalized treatment planning and health/wellness management solutions collectively represent a remaining 36% of application share, offering tailored insights for preventive care and lifestyle adjustments.
Hospitals and clinics represent the leading end‑user segment with approximately 41% of digital biomarker implementation due to their need for continuous patient data to inform acute and chronic care pathways, while pharmaceutical and biotechnology companies account for 33% of usage focused on clinical research enhancement; diagnostic laboratories and consumer health technology firms comprise the remaining 26% share. Within the fastest‑growing segment, adoption among research institutes is accelerating at a substantial pace as translational science programs integrate advanced biomarker technologies to support early‑stage discovery and predictive modeling, reflecting expanding research portfolios and computational capacity. Other end‑users such as telehealth service providers and wellness platform integrators contribute meaningfully to market breadth, with many reporting adoption rates in excess of 22% within specific regional markets.
North America accounted for the largest market share at 41% in 2025; however, Asia-Pacific is expected to register the fastest growth, expanding at a CAGR of 14.2% between 2026 and 2033.
North America leads due to high integration of wearable devices, AI-enabled analytics, and telehealth platforms, with over 52 million digital biomarker-enabled devices in use across hospitals and research centers. Europe follows with 28% market share, driven by Germany, the UK, and France, where regulatory compliance and explainable AI adoption are strong. Asia-Pacific reported 19% share in 2025, with China, India, and Japan leading demand through mobile health applications and advanced sensor infrastructure. South America and the Middle East & Africa collectively contribute 12% of the market, supported by increasing government initiatives, pilot telemedicine projects, and localized healthcare programs targeting chronic disease monitoring. The region-wise distribution highlights adoption intensity, infrastructure investment, and technology integration as primary factors shaping market growth across geographies.
How is healthcare innovation shaping continuous monitoring in North America?
North America holds 41% of the digital biomarker market share, led by hospitals, clinical research organizations, and pharmaceutical firms driving adoption. Regulatory frameworks, such as FDA digital health guidelines, and government incentives for telehealth deployment have accelerated technology integration. Technological advancements include AI-driven analytics, wearable devices, and cloud-based monitoring platforms. Local players like AliveCor have implemented AI-enabled cardiac monitoring solutions across 2,500 clinics, improving early arrhythmia detection by 37%. Consumer adoption is high, with enterprise healthcare and finance sectors integrating real-time monitoring, while patient behavior favors wearable and mobile health platforms. These dynamics reflect strong infrastructure support, early technology adoption, and regional emphasis on digital health transformation.
How are regulatory standards influencing digital health adoption in Europe?
Europe commands 28% of the digital biomarker market, with Germany, the UK, and France as leading contributors. The European Medicines Agency and GDPR compliance frameworks have increased demand for explainable, secure digital biomarker solutions. Adoption of emerging technologies like AI-enabled analytics and multimodal wearable devices is rising, particularly in clinical trial optimization. Local player Bioclinica has deployed remote patient monitoring platforms across multiple EU hospitals, enhancing chronic disease patient tracking by 33%. Regulatory pressure shapes consumer behavior, prioritizing solutions with clear data governance and transparency. Healthcare institutions in Europe increasingly integrate digital biomarkers to comply with stringent patient privacy and medical data standards.
What factors are accelerating digital health adoption in Asia-Pacific?
Asia-Pacific holds 19% of the digital biomarker market volume, with China, India, and Japan as top-consuming countries. Growth is supported by expanding mobile AI applications, telemedicine platforms, and high-volume wearable device manufacturing. Innovation hubs in Japan and Singapore focus on continuous sensor technology and AI-powered analytics for predictive healthcare. Local companies like iHealth Labs have scaled mobile-connected devices in hospitals and home monitoring programs, reaching over 8 million users in 2025. Consumer behavior favors mobile-first health solutions, with strong adoption in urban centers and e-commerce health platforms driving regional expansion.
How are localized healthcare initiatives driving digital biomarker adoption?
South America accounts for approximately 7% of the global digital biomarker market, with Brazil and Argentina leading deployment. Government programs incentivizing telemedicine and chronic disease monitoring have catalyzed infrastructure development in urban and semi-urban areas. Companies like Dasa in Brazil are implementing remote patient monitoring solutions for cardiovascular and metabolic health, improving engagement by 29%. Regional adoption reflects consumer preference for localized, language-adapted platforms and digital health education programs. Investment in hospital IT infrastructure and public health initiatives further support market expansion.
What is driving healthcare digitization in emerging markets of the Middle East & Africa?
The Middle East & Africa contributes 5% to the global digital biomarker market, with UAE and South Africa as major growth hubs. Regional trends include adoption in hospitals, oil & gas employee wellness programs, and telehealth networks. Technological modernization encompasses wearable sensors and AI-enabled monitoring platforms, while trade partnerships facilitate cross-border technology integration. Local players like LifePharma in UAE have deployed AI-assisted remote monitoring systems for chronic disease management, improving adherence by 24%. Consumers increasingly adopt wearable solutions for preventive care, with enterprises integrating digital biomarker platforms for workforce health monitoring.
United States: 41% market share; dominance driven by high production capacity, extensive clinical trial adoption, and advanced healthcare infrastructure.
Germany: 12% market share; leadership supported by regulatory compliance, explainable AI adoption, and high enterprise utilization in hospitals and research institutes.
The competitive environment in the Digital Biomarker market is dynamic and increasingly strategic, with an estimated 40+ active competitors globally integrating innovative analytics, wearable sensor technologies, and clinical validation platforms to capture market opportunities. The market shows a moderately fragmented structure, with a combined share of the top 5 companies approximating 45%, reflecting a landscape where neither a single player dominates entirely but alliance and differentiation are critical. Key players range from established med‑tech and pharmaceutical innovators to specialized digital health startups focused on niche biomarker solutions such as neurological, cardiovascular, and metabolic monitoring.
Strategic initiatives are shaping competition: partnerships between tech and life science firms are accelerating integrated platform development, while targeted product launches of AI‑enabled analytics modules and continuous monitoring devices enhance clinical value propositions. Mergers and acquisitions are increasingly evident, with larger players securing analytics specialists and device manufacturers to broaden portfolios. Innovation trends such as cloud‑native digital biomarker API ecosystems and federated learning frameworks improve interoperability and privacy compliance, influencing competitive positioning. Cross‑sector collaborations — for example, between analytics firms and contract research organizations — enhance market reach and validation capabilities. As decision‑makers assess competitive threats, differentiation through evidence‑based validation, regulatory alignment, and user‑centric design remains a key factor in securing enterprise and clinical adoption.
Evidation Health
Biogen
Koneksa Health
BioSensics
AliveCor, Inc.
Eyenuk, Inc.
Quibim
The Digital Biomarker market is being reshaped by advancements in sensor technologies, artificial intelligence, edge computing, and data interoperability standards that enable richer, more continuous health insights. Next‑generation wearable sensors now support multimodal data capture, allowing simultaneous collection of physiological (heart rate, skin temperature), behavioral (activity, sleep patterns), and contextual signals with sub‑second precision. Over 62% of new digital health deployments incorporate advanced inertial measurement units (IMUs) and optical photoplethysmography (PPG) sensors that improve signal fidelity and reduce noise in clinical settings. Artificial intelligence and machine learning algorithms are central to translating raw digital signals into actionable biomarker data. Contemporary deep learning models applied to time‑series health data can detect subtle anomalies—such as arrhythmia or respiratory irregularities—with accuracy improvements of 25–30% compared to traditional statistical approaches. These AI models increasingly leverage federated learning frameworks, enabling secure cross‑institutional model training without centralized data pooling, a capability that more than 45% of enterprise digital health platforms are integrating to meet privacy and compliance requirements.
Edge computing is another transformative enabler; local processing of biometric signals on devices reduces latency and bandwidth utilization, supporting real‑time alerts for clinicians and patients. Roughly 38% of wearable solutions now embed on‑device analytics to deliver immediate insights without continuous cloud connectivity. Interoperability standards, including FHIR (Fast Healthcare Interoperability Resources), are accelerating platform integration across electronic health records (EHRs), clinical trial management systems, and telehealth applications, promoting seamless data exchange across enterprise ecosystems. Emerging technologies such as digital twin modeling and predictive health meshes are also gaining traction. Digital twins—virtual representations of individual health profiles constructed from continuous digital biomarker feeds—can simulate disease progression and treatment response, enhancing personalized care planning. Meanwhile, predictive health meshes aggregate distributed biometric data with environmental and lifestyle indicators to provide contextualized risk assessments, improving precision in preventive health strategies. Collectively, these technological innovations are redefining how digital biomarkers are captured, analyzed, and operationalized across healthcare delivery and research domains.
• In December 2024, electronRx announced the launch of purpleDx, a cardiopulmonary assessment app compliant with medical device standards, enabling chronic respiratory disease patients to monitor digital lung function biomarkers at home and provide clinicians with real‑time patient data for tailored treatment plans at CES 2025. (telecommunicationspressreleases.com)
• In March 2025, the U.S. FDA granted 510(k) registration and listing for Scienza Health’s DigitalHumanOS™ platform, marking the first AI‑powered Digital Human system authorized as a Class II medical device with demonstrated 94% diagnostic accuracy in clinical interactions. (Scienza Health)
• In June 2025, IBM and Roche jointly introduced an AI‑enabled predictive glucose monitoring solution integrated with continuous glucose monitoring sensors, delivering advanced forecasting features for diabetic care that can anticipate hypoglycemic events up to 30 minutes before onset. (IBM Newsroom)
• In August 2025, Artera achieved U.S. FDA De Novo marketing authorization for its ArteraAI Prostate software, the first AI‑driven digital pathology tool authorized to prognosticate long‑term outcomes for non‑metastatic prostate cancer patients, establishing a new regulatory product category.
The scope of the Digital Biomarker Market Report encompasses a comprehensive assessment of digital biomarker ecosystems across technology, applications, user segments, and geographical regions. The report dissects system components such as data collection tools, data integration platforms, wearable sensors, mobile applications, and biosensors, quantifying their penetration and adoption patterns. It also examines the role of digital biomarkers in diverse clinical practices, including diagnostic monitoring, predictive and prognostic biomarker applications, and their deployment across therapeutic areas such as cardiovascular, neurodegenerative, metabolic, respiratory, and chronic disease management.
Geographic analysis spans North America, Europe, Asia‑Pacific, South America, and Middle East & Africa, profiling how regional infrastructure, regulatory dynamics, and consumer behavior shape adoption. For example, North America’s advanced healthcare IT infrastructure drives high integration of continuous monitoring platforms, while Asia‑Pacific shows rapid mobile health app adoption and scalable wearable deployment. The report also articulates key end‑users, including hospitals and clinics, pharmaceutical and biotech firms, clinical research organizations, and digital health service providers, emphasizing the decision criteria and utilization intensity of each segment.
Emerging niches—such as decentralized clinical trials using digital endpoints, AI‑augmented pathology analysis, and real‑time remote patient monitoring—are featured with metric‑based insights on device utilization rates and technology integration trends. The coverage includes innovation vectors like multimodal data fusion, real‑time predictive analytics, and secure interoperable architectures enabling seamless exchange across electronic records and clinical systems. Overall, the report equips industry professionals with actionable analysis of adoption patterns, technological drivers, user demands, and strategic opportunities across the evolving global digital biomarker landscape.
| Report Attribute/Metric | Report Details |
|---|---|
|
Market Revenue in 2025 |
USD V2025 Million |
|
Market Revenue in 2033 |
USD V2033 Million |
|
CAGR (2026 - 2033) |
82.5% |
|
Base Year |
2025 |
|
Forecast Period |
2026 - 2033 |
|
Historic Period |
2021 - 2025 |
|
Segments Covered |
By Types
By Application
By End-User
|
|
Key Report Deliverable |
Revenue Forecast, Growth Trends, Market Dynamics, Segmental Overview, Regional and Country-wise Analysis, Competition Landscape |
|
Region Covered |
North America, Europe, Asia-Pacific, South America, Middle East, Africa |
|
Key Players Analyzed |
Philips Healthcare website, IBM Watson Health overview, Medtronic digital health solutions, Evidation Health, Biogen, Koneksa Health, BioSensics, AliveCor, Inc., Eyenuk, Inc., Quibim |
|
Customization & Pricing |
Available on Request (10% Customization is Free) |
